Summary
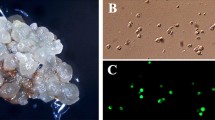
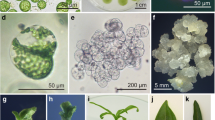
A system to regenerate fertile rice (Oryza sativa L.) plants (both indica and japonica varieties) from protoplasts isolated from anther-derived embryogenic haploid suspension cultures has been established. Green plants were regenerated from protoplast-derived cell clusters five months after suspension culture initiation. Protoplast yields and subsequent growth of the protoplast-derived microcalli were enhanced by transferring suspension cells into AA medium (Muller et al. 1978) three to four days prior to protoplast isolation. Protoplasts were cultured initially in Kao medium (Kao et al. 1977) and in association with nurse cells for four weeks. Protoplast-derived microcalli were transferred onto N6 (Chu et al. 1975) or MS (Murashige and Skoog 1962) media for callus proliferation. Callus growth was more rapid and the calli were more enbryogenic when grown on N6 medium. The 2,4-D concentration used to develop the suspension culture was important. Cell cultures grown in medium containing 0.5 mg/l 2,4-D released protoplasts whose plating efficiency was higher than for protoplasts obtained from suspension cultures grown in 2.0 mg/l 2,4-D. However, suspension cells grown in 2.0 mg/l 2,4-D were superior with regard to the ability of protoplast-derived calli to regenerate green plants. Amongst several hormone treatments evaluated, a combination of 0.5 mg/l NAA + 5.0 mg/l BAP resulted in the largest number of green plants regenerated. There were no significant differences between BAP or kinetin regarding total number of plants regenerated. More than 200 green plants have been produced form six independently initiated suspension cell lines. The number of regenerated plants per 106 protoplats plated anged from 0.4 to 20.0, and the average seed fertility of single panicles of these RO plants was about 40%.
Similar content being viewed by others
References
Abdullah R., Cocking E.C., Thompson J.A. (1986) Bio/Technol 4: 1087–1090.
Chu C.C., Wang C.C., Sun C.S., Hsu C., Yin K.C., Chu C.Y., and Bin F.Y. (1975) Scientia Sinica 18: 659–668.
Coulibaly M.Y., Demarly Y. (1986) Z. Pflanzenzuchtg. 96: 79–81.
Datta S.K., Peterhans A., Datta K. and Potrykus I. (1990) Bio/Technology 8: 736–740.
Fujimura T., Sakurai M., Akagi H., Negishi T., Hirose A. (1985) Plant Tissue Culture Lett. 2: 74–75.
Gamborg, O.L., Miller, R.A., Ojima, K. (1968) Exp. Cell Res. 50: 151–158.
Hodges T. K., Peng J., Lyznik L.A. and Koetje D.S. (1991) Rice Biotechnology, eds. Toenniessen G. and Khush G. pp. 157–175
Kyozuka I, Hayashi Y., Shimamoto K. (1987) Mol. Gen Genet 206: 408–413.
Kao K.N. (1977) Mol. Gen. Genet. 150: 225–230.
Koetje D.S., Grimes H.D., Wang Y-C, Hodges T.K. (1989) J Plant Physiol. 135: 184–190.
Lee L., Schroll R.E., Grimes H.D., Hodges T.K. (1989) Planta 178: 325–333.
Linsmaier E.M. and Skoog F. (1965) Physiologia Plantarum 18: 100–127.
Murashige T and Skoog F. (1962) Physiologia Plantarum 15: 473–497.
Muller AJ. and Grafe R. (1978) Mol. Gen. Genet. 161: 67–76.
Niizeki H. and Oono K. (1968) Proc. Jpn. Acad. 44: 554–557.
Nishi T., Yamada Y., Takahashi E. (1968) Nature 219: 508–509.
Peng J., Kononowicz H., and Hodges T.K. (1992) Theor. Appl. Genet. 83: 855–865.
Shimamoto K., Terada R., Izawa T., Fujimoto H. (1989) Nature 338: 274–276.
Thompson J.A., Abdullah R., Cocking E.C. (1986) Plant Sci. 47: 123–134.
Toriyama K., Hinata K. (1985) Plant Sci. 41: 179–183.
Toriyama K., Hinata K., Sasaki T. (1986) Theor. Appl. Genet. 73: 16–19.
Yamada Y., Yang Z-Q., Tang D-T. (1986) Plant Cell Rep. 5: 85–88.
Author information
Authors and Affiliations
Additional information
Communicated by A. R. Gould
Rights and permissions
About this article
Cite this article
Su, RC., Rudert, M.L. & Hodges, T.K. Fertile indica and japonica rice plants regenerated from protoplasts isolated from embryogenic haploid suspension cultures. Plant Cell Reports 12, 45–49 (1992). https://doi.org/10.1007/BF00232422
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00232422