Summary
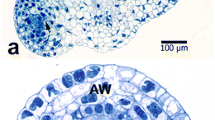
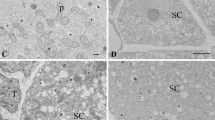
The pattern of degeneration in mature synergids of pearl millet prior to pollination was examined by transmission electron microscopy after conventional and antimonate fixation to precipitate loosely sequestered calcium (Ca). The extent of degeneration and the distribution of Ca in a synergid varied among samples, and also between some sister synergids. However, there seemed to be no difference between sister synergids in the total amount of precipitates present in each cell. Characteristic signs of degeneration in the antimonatefixed synergids were: a gap above the filiform apparatus with fibrillar material, precipitates, and fusing or disintegrating membranes; increased precipitates in the nucleus, nucleolus, and endoplasmic reticulum; fusion or collapse of a few vacuoles in the chalazal core with precipitates and flocculent material deposited nearby. The numerous mitochondria and proplastids in the micropylar portion of the synergid remained intact and mostly free of precipitates. The shape and content of the chalazal vacuoles appeared to be disparate and dependent on the fixation procedure as well as the extent of degeneration within a synergid. The results suggest that the sister synergids of pearl millet undergo autonomous degeneration apparently in two independent sequences that may be spatially and temporally separated. The vacuoles appear to be dynamic organelles that store Ca in association with some other material. A high concentration of Ca may be localized along or between the common wall of the two filiform apparatuses.
Similar content being viewed by others
References
Chaubal R, Reger BJ (1990) Relatively high calcium is localized in synergid cells of wheat ovaries. Sex Plant Reprod 3:98–102
Chaubal R, Reger BJ (1992a) alcium in the synergid cells and other regions of pearl millet ovaries. Sex Plant Reprod 5:34–46
Chaubal R, Reger BJ (1992b) The dynamics of calcium distribution in the synergid cells of wheat following pollination. Sex Plant Reprod 5:206–213
Dujardin M, Hanna W (1984) Microsporogenesis, reproductive behavior, and fertility in five Pennisetum species. Theor Appl Genet 67:197–201
Dute RR, Peterson CM, Rushing AE (1989) Ultrastructural changes of the egg apparatus with fertilization and proembryo development of soybean Glycine max (Fabaceae). Ann Bot 64:123–135
Folsom MW, Cass DD (1992) Embryo sac development in soybean: the central cell and aspects of fertilization. Am J Bot 79:1407–1417
He C-P and Yang H-Y (1992) Ultracytochemical localization of calcium in the embryo sac of sunflower. Chin J Bot 4:99–106
Huang B-Q, Russell SD (1992a) Synergid degeneration in Nicotiana: a quantitative, fluorochromatic, and chlorotetracycline study. Sex Plant Reprod 5:151–155
Huang B-Q, Russell SD (1992b) Female germ unit: organization, reconstruction and isolation. Int Rev Cytol 140:233–293
Jensen WA (1965) The ultrastructure and histochemistry of the synergids of cotton. Am J Bot 52:238–256
Jensen WA, Fisher DB (1968) Cotton embryogenesis: the entrance and discharge of the pollen tube in the embryo sac. Planta 78:158–183
Jensen WA, Ashton ME, Beasley CA (1983) Pollen tube — embryo sac interaction in cotton. In: Mulcahy DL, Ottaviano E (eds) Pollen: biology and implications for plant breeding. Elsevier Biomedical, New York, pp 67–72
Mascarenhas JP and Machlis L (1964) Chemotropic response of the pollen of Antirrhinum majus to calcium. Plant Physiol 39:70–77
Mogensen HL (1978) Pollen tube-synergid interaction in Proboscidea louisianica (Martineaceae). Am J Bot 65:953–964
Mogensen HL, Suthar HK (1979) Ultrastructure of the egg apparatus of Nicotiana tabacum (Solanaceae) before and after fertilization. Bot Gaz 140:168–179
Narayanaswami S (1956) Structure and development of the caryopsis in some Indian millets. VI. Setaria italica. Bot Gaz 118:112–122
Pluijm JE van der (1964) An electron microscopic investigation of the filiform apparatus in the embryo sac of Torenia fournieri. In: Linskens HF (ed) Pollen physiology and fertilization. North Holland Pub Co, Amsterdam, pp 8–16
Reger BJ, Chaubal R, Pressey R (1992) Chemotropic responses by pearl millet pollen tubes. Sex Plant Reprod 5:47–56
Russell SD (1992) Double fertilization. Int Rev Cytol 140:357–388
Russell SD, Rougier M, Dumas C (1990) Organization of the early post-fertilization megagametophyte of Populus deltoides: ultrastructure and implications for male cytoplasmic transmission. Protoplasma 155:153–165
Simpson CE, Bashaw EC (1969) Cytology and reproductive characteristics in Pennisetum setaceum. Am J Bot 56:31–36
Tirlapur UK, Van Went JL, Cresti M (1993) Visualization of membrane calcium and calmodulin in embryo sacs in situ and isolated from Petunia hybrida L. and Nicotiana tabacum L. Ann of Bot 71:161–167
Van Went J, Cresti M (1988) Pre-fertilization degeneration of both synergids in Brassica campesteris ovules. Sex Plant Reprod 1:208–216
Wilms HJ (1981) Pollen tube penetration and fertilization in spinach. Acta Bot Neerl 30:101–122
You R, Jensen WA (1985) Ultrastructural observations of the mature megagametophyte and the fertilization in wheat (Triticum aestivum). Can J Bot 63:163–178
Author information
Authors and Affiliations
Additional information
On specific Cooperative Agreement 58-6612-8-002 with the Department of Biochemistry, University of Georgia, Athens, GA 30602, USA
Rights and permissions
About this article
Cite this article
Chaubal, R., Reger, B.J. Prepollination degeneration in mature synergids of pearl millet: an examination using antimonate fixation to localize calcium. Sexual Plant Reprod 6, 225–238 (1993). https://doi.org/10.1007/BF00231899
Issue Date:
DOI: https://doi.org/10.1007/BF00231899