Summary
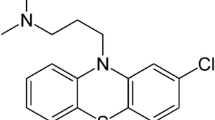
Calcineurin was dicovered as an inhibitor of calmodulin stimulated cyclic AMP phosphodiesterase and its ability to act as a calmodulin binding protein largely explains its inhibitory action on calmodulin regulated enzymes. Recent studies establish calcineurin as the enzyme protein phosphatase whose activity is regulated by calmodulin and a variety of divalent metals. In this work, we have investigated the effects of several agents including sulfhydryl agents, trifluoperazine (a calmodulin antagonist), PPi, NaF and orthovanadate and of tryptic proteolysis on the calcineurin inhibition of cyclic AMP phosphodiesterase (called inhibitory activity) and on protein phosphatase activity. Inhibitors for sulfhydryl groups (pHMB, NEM) inhibited phosphatase activity without any effect on the inhibitory activity. Dithioerythritol completely reversed the inhibition by pHMB. Limited proteolysis of calcineurin caused an activation of basal phosphatase activity with a complete loss of inhibitory activity. Phosphatase activity of the proteolyzed calcineurin was not stimulated by calmodulin. The presence of calmodulin along with calcineurin during tryptic digestion appeared to preserve the stimulation of phosphatase by Ca2+-calmodulin. [3H]-Trifluoperazine (TFP) was found to be incorporated irreversibly into calcineurin in the presence of ultraviolet light. This incorporation was evident into the A and B subunits of calcineurin. TFP-caused a decrease in the phosphatase activity and an increase in its inhibitory activity. [3H]-TFP incorporation into the A subunit was drastically decreased in the proteolyzed calcineurin. This was also true when the [3H]-TFP incorporated calcineurin was subjected to tryptic proteolysis. The incorporation into the B unit was essentially unaffected in the trypsinized calcineurin. Phosphatase activity was inhibited by orthovanadate, NaF, PPi, and EDTA. Inhibitions by these compounds were more pronounced when the phosphatase was determined in the presence of Ca2+-cahnodulin than in their absence.
Similar content being viewed by others
References
Klee CB, Draetta GF, Hubbard MJ: Calcineurin. In: A Meisler (ed.) Advan Enzymol Wiley Interscience, New York, 1988, pp 149–200
Klee CB, Crouch TH, Krinks MH: Calcineurin: a calcium- and calmodulin-binding protein of the nervous system. Proc Natl Acad Sci USA 76: 6270–6273, 1979
Sharma R, Desai R, Waisman DM, Wang JH: Purification and subunit structure of bovine brain modulator binding protein. J Biol Chem 254: 4276–4282, 1979
Wallace RW, Lynch TJ, Tallant EA, Cheung WY: Purification and characterization of an inhibitor protein of brain adenylate cyclase and cyclic nucleotide phosphodiesterase. J Biol Chem 254: 377–382, 1979
Klee DB, Krinks NH: Purification and cyclic 3′,5′-nucleotide phosphodiesterase inhibitory protein by affinity chromatography on activator protein coupled to sepharose. Biochemistry 17: 120–126, 1978
Richman PG, Klee CB: Interaction of 125I-labeled Ca2+-dependent regulator proteins with cyclic nucleotide phosphodiesterase and its inhibitory proteins. J Biol Chem 253: 6323–6326, 1978
Sharma RK, Desai R, Thompson TR, Wang JH: Purification of the heatstable inhibitor of the Ca2+-activated cyclic nucleotide phosphodiesterase by affinity chromatography. Can J Biochem 56: 598–604, 1978
Stewart AA, Ingebritsen TS, Manalan A, Klee CB, Cohen P: Discovery of a Ca2+- and calmodulin-dependent protein phosphatase. Probable identity with calcineurin (CaMBP80). FEBS Lett 137: 80–84, 1982
Yang SD, Tallant EA, Cheung WY: Calcineurin is a calmodulin-dependent protein phosphatase. Biochem Biophys Res Commun 106: 1419–1425, 1982
Pallen CJ, Wang JH: Calmodulin stimulated dephosphorylation of p-nitrophenyl phosphate and free phosphotyrosine by calcineurin. J Biol Chem 258: 8550–8553, 1983
Pallen CJ, Wang JH: Regulation of calcineurin by metal ions. Mechanism of activation by Ni2+ and an enhanced response to Ca2−/calmodulin. J Biol Chem 259: 6134–6141, 1984
Gupta RC, Khandelwal RL, Sulakhe PV: Intrinsic phosphatase activity of bovine brain calcineurin requires a tightly bound trace metal. FEBS Lett 169: 251–255, 1984
Gupta RC, Khandelwal RL, Sulakhe PV: Isolation and characterization of calcineurin from bovine brain. Can J Physiol Pharmacol 63: 1000–1006, 1985
Winkler MA, Merat DL, Tallant EA, Hawkins S, Cheung WY: Catalytic site of calmodulin-dependent protein phosphatase from bovine brain resides in subunit A. Proc Natl Acad Sci USA 81: 3054–3058, 1984
Gupta RC, Khandelwal RL, Sulakhe PV: Resolution of bovine brain calcineurin subunits: Stimulatory effect of subunit B on subunit A phosphatase activity. FEBS Lett 190: 104–108, 1985
Gupta RC, Khandelwal RL, Sulakhe PV: Interaction amongst calcineurin subunits: Stimulatory and inhibitory effects of subunit B on calmodulin stimulation of subunit A phosphatase activity depend on Mn2− exposure of the holoenzyme prior to its dissociation by urea. FEBS Lett 196: 39–43, 1986
Sharma RK, Wang TH, Wirch E, Wang JH: Purification and properties of bovine brain calmodulin-dependent cyclic nucleotide phosphodiesterase. J Biol Chem 255: 5915–5923, 1980
Taussky HH, Shorr E: A microcolorimetric method for the determination of inorganic phosphorus. J Biol Chem 202: 675–685, 1953
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ: Protein measurement with the Folin phenol reagent. J Biol Chem 193: 265–275, 1951
Aitken A, Klee CB, Cohen P: The structure of the B subunit of calcineurin. Eur J Biochem 139: 663–671, 1984
Watterson DM, Sharief F, Vanaman TC: The complete amino acid sequence of the Ca2+-dependent modulator protein (calmodulin) of bovine brain. J Biol Chem 255: 962–975, 1980
Levin RM, Weiss B: Binding of trifluoperazine to the calcium-dependent activator of cyclic necleotide phosphodiesterase. Mol Pharmacol 13: 690–697, 1977
Tonks NK, Cohen P: Calcineurin is a calcium ion-dependent, calmodulin-stimulated protein phosphatase. Biochim Biophys Acta 747: 191–193, 1983
Wang JH, Sharma RK, Tam SW: Calmodulin-binding proteins. In: WY Cheung (ed.) Calcium and Cell Function. Academic Press, New York, 1980, Vol 1, pp 305–328
Pallen CJ, Wang JH: Calcineurin-phosphatase: Ni2+-binding properties. Fed Proc 44: 707, 1985
King MM: Modification of the calmodulin-cumulated phosphatase, calcineurin by sulfhydryl reagents. J Biol Chem 261: 4081–4084, 1986
Levin RM, Weiss B: Selective binding of antipsychotics and other psychoactive agents to the calcium-dependent activator of cyclic nucleotide phosphodiesterase. J Pharmacol Exp Ther 208: 454–459, 1979
Weiss B, Levin RM: Mechanism for selectively inhibiting the activation of cyclic nucleotide phosphodiesterase and adenylate cyclase by antipsychotic agents. Adv Cyclic Nucleotide Res 9: 285–303, 1978
Manalan AS, Klee CB: Activation of calcineurin by limited proteolysis. Proc Natl Acad Sci USA 80: 4291–4295, 1983
Tallant EA, Cheung WY: Activation of bovine brain calmodulin-dependent protein phosphatase by limited trypsinization. Biochemistry 23: 973–979, 1984
Li H-C: Phosphoprotein phosphatases. Curr Top Cell Regul 21: 129–174, 1982
Hsiao KJ, Sandberg AR, Li HC: The role of ATP and divalent cations in the regulation of a cardiac phosphorylase phosphatase (phosphoprotein phosphatase) of Mr = 35,000. J Biol Chem 253: 6901–6907, 1978
Khatra BS, Soderling TR: Reversible inhibition of skeletal muscle phosphoprotein phosphatase by ATP, phosphate and fluoride. Biochem Biophys Res Commun 85: 647–654, 1978
Mackenzie CW III, Bulbulian GJ, Bishop JS: Use of fluoride to inactivate phosphorylase a phosphatases from rat liver cytosol. Presence of fluoride-insensitive glycogen synthase-specific phosphatase. Biochim Biophys Acta 614: 413–424, 1980
Goris J, Pijnenborg-Vercruysse L, Merlevede W: Effect of fluoride on liver phosphorylase phosphatase. Biochim Biophys Acta 268: 158–165, 1972
Yan SC, Graves DJ: Inactivation and reactivation of phosphoprotein phosphatase. Molec Cell Biochem 42: 21–29, 1982
Shacter-Noiman E, Chock PB: Properties of a Mr = 38,000 phosphoprotein phosphatase. Modulation by divalent cations, ATP, and fluoride. J Biol Chem 258: 4214–4219, 1983
Gupta RC, Khandelwal RL, Sulakhe PV: Divalent cation effects on calcineurin phosphatase: Differential involvement of hydrophobic and metal binding domains in the regulation of the enzyme activity. (accompanying paper)
Laemmli UK: Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature (London) 227: 680–685, 1970
Bonner WM, Laskey RA: A film detection method for tritium-labeled proteins and nucleic acids in polyacrylamide gels. Eur J Biochem 46: 83–88, 1974
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gupta, R.C., Khandelwal, R.L. & Sulakhe, P.V. Effects of sulfhydryl agents, trifluoperazine, phosphatase inhibitors and tryptic proteolysis on calcineurin isolated from bovine cerebral cortex. Mol Cell Biochem 97, 43–52 (1990). https://doi.org/10.1007/BF00231700
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00231700