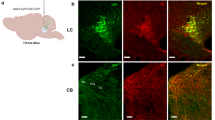
Summary
Multi-unit neuronal activity was recorded in posterior cingulate cortex (area 29) and the anterior ventral (AV) thalamic nucleus during discriminative instrumental avoidance learning wherein a response (stepping in an activity wheel) to a 0.5-s tone (CS+) prevented a foot-shock 5 s after CS+ onset. Presentations of a different tone (CS-) on 50% of the conditioning trials in an irregular sequence with the CS+ did not predict shock and thus required no response. Two groups of rabbits received intracranial micro-injections of 6-hydroxydopamine (6-OHDA) to locally deplete the NE in area 29 or the AV nucleus. Vehicle was injected in the non-depleted area in each group and a third group received vehicle injections in both areas. Dopamine neurons in subjects that received 6-OHDA were protected by pre-treatment with GBR-12909. Neuronal data were collected during two pre-training sessions in response to the tones only and when the tones and shock were presented unpaired. Thalamically depleted rabbits made more, and cortically depleted rabbits made fewer, avoidance responses than controls during the early stages of behavioral acquisition, and cortically depleted rabbits made fewer responses than controls and thalamically depleted rabbits during extinction testing administered after the completion of acquisition. One effect of NE depletion on neuronal activity was entirely local: elimination of neuronal sensitization effects (enhanced discharges elicited by tones during the unpaired tone-shock pre-training treatment relative to pre-training with tones only). Other neuronal effects of NE depletion were system-wide, i.e., they occurred whether the depletion was cortical or thalamic. These were: attenuation of area 29 tone-elicited neuronal discharges and enhancement of AV thalamic discharges before and during training; elimination in area 29 of neuronal discrimination between CS+ and CS-, induced in controls by CS+-shock pairings in the first conditioning session; induction of this neuronal discrimination, not present in controls, in the AV nucleus during the first conditioning session; attenuation of discharge enhancements elicited in controls by unexpected stimuli (presentation of auditory stimuli different in quality and incidence from the CS+). Excepting the noted losses at the outset of training, the results did not support an involvement of NE in the production of cingulate cortical or AV thalamic excitatory and discriminative training-induced neuronal activity. The system-wide alterations due to NE depletion implicated NE in the processing of unexpected events and in the production of dynamic neuronal patterns relevant to mnemonic retrieval. Several of the depletion-related neuronal changes were similar to the effects of hippocampal formation (subicular) lesions, suggesting that NE-dependent functions in area 29 and the AV nucleus are governed by hippocampal efferents, which may control the release of NE in these areas.
Similar content being viewed by others
References
Abercrombie ED, Zigmond MJ (1989) Partial injury to central noradrenergic neurons: reduction of tissue norepinephrine content is greater than reduction of extracellular norepinephrine measured by microdialysis. J Neurosci 9:4062–4067
Archer T, Mohammed AK, Ross SB, Soderberg U (1983) T-maze learning, spontaneous activity and food intake in recovery following systemic administration of the noradrenaline neurotoxin DSP4. Pharmacol Biochem Behav 19:121–130
Aston-Jones G (1985) Behavioral functions of locus coeruleus derived from cellular attributes. Physiol Psychol 13:118–126
Aston-Jones G, Bloom FE (1981) Norepinephrine containing locus coeruleus neurons in behaving rats exhibited pronounced responses to non-noxious environmental stimuli. J Neurosci 1:887–900
Aston-Jones G, Ennis M, Pieribone VA, Nickell WT, Shipley MT (1986) The brain nucleus locus coeruleus: restricted afferent control of a broad efferent network. Science 234:734–737
Bonanno G, Raiteri M (1987) A carrier for GABA uptake exists on noradrenaline nerve endings in selective rat brain areas but not on serotonin terminals J Neurotrans 69:59–70
Brogden WJ, Culler FA (1936) A device for motor conditioning of small animals. Science 83:269
Buchwald J, Holstein SB, Weber DS (1973) Multiple-unit recording: technique, interpretation and experimental applications. In:Thompson RF, Patterson MM (eds) Bioelectric Recording Techniques: Part A. Cellular processes and brain potentials. Academic Press, New York, pp 242
Crow TJ (1968) Cortical synapses and reinforcement: a hypothesis, Nature 219:736–737
Dahl D, Sarvey JM (1989) Norepinephrine induces pathway-specific long-lasting potentiation and depression in the hippocampal dentate gyrus. Proc Natl Acad Sci 86:4776–4780
Foote SL, Freedman R, Oliver AP (1975) Effects of putative neurotransmitters on neuronal activity in monkey auditory cortex. Brain Res 86:229–242
Fox CA, Eichman JA (1959) A rapid method for locating intracerebral electrode tracks. Stain Technol 34:39–42
Gage FH, Bjorklund A, Stenevi U (1983) Local regulation of compensatory noradrenergic hyperactivity in the partially denervated hippocampus. Nature 303:819–821
Gabriel M (1967) Intersession delay and avoidance extinction to training and generalization stimuli. Psychon Sci 9:243–244
Gabriel M (1990) Functions of anterior and posterior cingulate cortex during avoidance learning in rabbits. Prog Brain Res 85:467–483
Gabriel M, Vogt J (1970) Incubation of avoidance CRs in the rabbit produced by increase over time in stimulus generalization to apparatus stimuli. Behav Neur Biol 7:113–125
Gabriel M, Saltwick SE (1977) Effects of unpaired footshock on rabbit limbic and auditory neuronal responses to tone stimuli. Physiol Behav 19:29–34
Gabriel M, Saltwick SE, Miller JD (1980) Interaction of the laminae of cingulate cortex and the anteroventral thalamus during behavioral learning. Science 208:1050–1052
Gabriel M, Lambert RW, Foster K, Orona E, Sparenborg S, Maiorca R (1983) Anterior thalamic lesions and neuronal activity in the cingulate and retrosplenial cortices during discriminative avoidance behavior in rabbits. Behav Neurosci 97:675–696
Gabriel M, Sparenborg S, Stolar N (1987) Hippocampal control of cingulate cortical and anterior thalamic information processing during learning in rabbits. Exp Brain Res 67:131–152
Gabriel M, Poremba A, Ellison-Perrine C, Miller JD (1990) Brainstem mediation of learning and memory. In:Klemm WR, Vertes RP (eds) Brainstem mechanisms of behavior. Wiley, New York, pp 269–314
Gabriel M, Kubota Y, Sparenborg S, Straube K, Vogt B (1991a) Effects of cingulate cortical lesions on avoidance learning and training-induced unit activity in rabbits. Exp Brain Res 86:585–600
Gabriel M, Vogt BA, Kubota Y, Poremba A, Kang E (1991b) Training-stage related neuronal plasticity in limbic thalamus and cingulate cortex during learning in rabbits: a possible key to mnemonic retrieval Behav Brain Res 46:175–185
Gillespie D, Manier DH, Sanders-Bush E, Sulser F (1988) The serotonin-norepinephrine link in brain. II. Role of serotonin in the regulation of beta-adrenoceptors in the low agonist affinity conformation. J Pharmacol Exp Ther 244:154–159
Girgis M, Shih-Chang W (1981) A new stereotaxic atlas of the rabbit brain. Green, St. Louis
Gray JA (1984) The neuropsychiology of anxiety: an enquiry into the functions of the septo-hippocampal system. Oxford University Press, Oxford
Harley CW (1987) A role for norepinephrine in arousal emotion and learning? Limbic modulation by norepinephrine and the Kety hypothesis. Prog Neuropsychopharm Biol Psychiat 11:419–458
Heikkila RE, Manzino L (1984) Behavioral properties of GBR1 2909, GBR-13069 and GBR-13098: specific inhibitors of dopamine uptake. Eur J Pharmacol 103:241–248
Isaacson RA (1982) The limbic system, 2nd ed. Plenum Press, New York
Jaffard R, Beracochea D, Cho Y (1991) The hippocampalmammillary system: anterograde and retrograde amnesia. Hippocampus 1:275–278
Jones EG, Moore RY (1977) Ascending projections of the locus coeruleus in the rat. 2. Autoradiographic study. Brain Res 127:23–53
Kandel ER, Swartz JH (1984) Molecular biology of learning: modulation of transmitter release. Science 218:433–443
Kang E, Kubota Y, Poremba A and Gabriel M (1990) Hippocampal lesions, limbic cortical and thalamic training-induced unit activity, avoidance conditioning and response to different forms of novelty in rabbits. Soc Neurosci Abstr Bull 113. 19:264
Kasamatsu T, Heggelund P (1982) Single cell responses in cat visual cortex to visual stimulation during iontophoresis of noradrenaline. Exp Brain Res 45:317–327
Kety SS (1970) The biogenic amines in the central nervous system: their possible roles in arousal, emotion and learning. In:Schmitt FO (ed) The neuroscience second study program. Rockefeller University Press, New York, pp 324–335
Kubota Y, Gabriel M (1992) Anterodorsal thalamic nuclear lesions and training-induced neuronal activity in the anterior ventral thalamic nucleus and posterior cingulate cortex during learning in rabbits. (submitted)
Magistretti PJ, Morrison JH (1988) Noradrenaline and vasoactive intestinal peptide-containing neuronal systems in neocortex: Functional convergence with contrasting morphology. Neuroscience 24:367–378
Marrocco RT, Lane RF, McClurkin JW, Blaha CD, Alkire MF (1987) Release of cortical catecholamines by visual stimulation requires activity in thalamocortical afferents of monkey and cat. J Neurosci 7:2756–2767
Mason ST, Fibiger HC (1978) Noradrenalin and spatial memory. Brain Res 156:352–386
Mignard M, Bentzinger D, Bender N, Gabriel M (1987) Type I and type II theta-like unit activity in structures of the Papez circuit during differential avoidance conditioning in rabbits. Soc Neurosci Abstr Bull 305. 15:1103
Mogensen GJ (1990) Brainstem systems for the control of behavioral acts. In:Klemm WR, Vertes RP (eds) Brainstem mechanisms of behavior. Wiley, New York, pp 171–198
Moises HC, Waterhouse BD, Woodward DJ (1981) Locus coeruleus stimulation potentiates Purkinje cell responses to afferent input: the climbing fiber system. Brain Res 222:43–64
Morrison JH, Grzanna R, Molliver ME, Coyle JT (1978) The distribution and orientation of noradrenergic fibers in neocortex of the rat: an immunofluorescence study. J Comp Neurol 181:17–40
Morrison JH, Molliver ME, Grzanna R, Coyle JT (1981) The intra-cortical trajectory of the coeruleo-cortical projection in the rat: a tangentially organized cortical afferent. Neuroscience 6:139–158
Pineda J, Foote S, Neville H (1989) Effects of locus coeruleus lesions on auditory, long-latency, event-related potentials in monkey. J Neurosci 9:81–93
Rescorla RA (1967) Pavlovian conditioning and its proper control procedures. Psychol Rev 74:71–80
Saller CF, Salama AI (1984) Rapid automated analysis of biogenic amines and their metabolites using reversed-phase high-performance liquid chromatography with electrochemical detection. J Chromatogr 309:287–298
Sara SJ (1991) Noradrenaline and memory: neuromodulatory influences on retrieval. In:Memory — neurochemical and abnormal perspectives. Harwood, New York, pp 105–128
Sara SJ, Segal M (1991) Plasticity of sensory responses of locus coeruleus neurons in the behaving rat: implications for cognition. Prog Brain Res 88:571–585
Segal M, Bloom F (1976) The action of noreinephrine in the rat hippocampus. III. Hippocampal cellular responses to locus coeruleus stimulation in the awake rat. Brain Res 107:499–512
Selden N, Robbins T, Everitt B (1990) Enhanced behavioral conditioning to context and impaired behavioral and neuroendocrine responses to conditioned stimuli following ceruleocortical noradrenergic lesions: support for an attentional hypothesis of central noradrenergic function. J Neurosci 10:531–539
Stolar N, Sparenborg S, Donchin E, Gabriel M (1989) Conditional stimulus probability and activity of hippocampal, cingulate cortical and limbic thalamic neurons during avoidance conditioning in rabbits. Behav Neurosci 103:919–934
Swanson LW, Hartman BK (1975) The central adrenergic system. An immunofluorescence study of the localization of cell bodies and their efferent connections in the rat utilizing dopamine-betahydroxylase as a marker. J Comp Neurol 163:467–506
Van Groen T, Wyss JM (1990a) The postsubicular cortex in the rat: characterization of the fourth region of the subicular complex and its connections. Brain Res 529:165–177
Van Groen T, Wyss JM (1990b) The connections of the presubiculum and parasubiculum of the rat. Brain Res 518:227–243
Waterhouse BD, Woodward DJ (1980) Noradrenergic modulation of somatosensory cortical neuronal responses to iontophoretically applied putative neurotransmitters. Exp Neurol 69:30–49
Waterhouse BD, Moises HC, Woodward DJ (1980) Interaction of norepinephrine with cerebrocortical activity evoked by stimulation of somatosensory afferent pathways in the rat. Exp Neurol 67:11–34
Waterhouse BD, Moises HC, Woodward DJ (1981) Alpha-receptor-mediated facilitation of somatosensory cortical neuronal responses to excitatory synaptic inputs and iontophoretically applied acetylcholine. Neuropharmacology 20:907–920
Winer BJ (1962) Statistical principles in experimental design. McGraw-Hill, New York
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sparenborg, S., Gabriel, M. Local norepinephrine depletion and learning-related neuronal activity in cingulate cortex and anterior thalamus of rabbits. Exp Brain Res 92, 267–285 (1992). https://doi.org/10.1007/BF00227970
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00227970