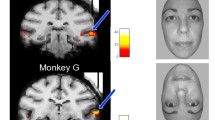
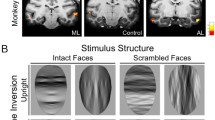
Abstract
The ability of a human observer to detect the presence of a briefly flashed picture of a face can depend on the picture's spatial configuration, that is on whether its features are rearranged (jumbled) or are in their normal configuration. The face-detection effect (FDE) is found under conditions of backward masking, when the presence of a face can be detected with shorter masking intervals when it is in the normal than when in the rearranged configuration. A similar effect is found when the subject is asked to classify the face as rearranged or not — the face-classification effect (FCE). Part of the interest of the FDE and the FCE is that they show how the configuration of a stimulus can be an important factor in the perceptual processing which leads to detection and classification of the stimulus. To analyse these effects we recorded from single neurons in the cortex in the superior temporal sulcus of macaques when they were shown (in a visual fixation task) normal and rearranged faces under backward masking conditions shown in experiments 2 and 3 to produce, with the same apparatus, the FCE, and also to produce comparable effects on the identification of which face was present (called hereafter the face-identification effect), and also of the clarity of the face. We found in experiment 1 that there are some face-selective neurons which respond to faces only, or better, when the features in the faces are in their normal configuration rather than rearranged. We also showed in this experiment that the difference in the response to the normal as compared to the rearranged faces became greater when the masking stimulus was delayed more. Thus, at intermediate delays, there are more neurons active for the normal than for the rearranged face. We therefore propose that the FDE, the FCE, and the face-identification effect arise because the total number of neurons activated by faces in their normal configuration is greater than that activated by rearranged faces, because of the sensitivity of some face-selective neurons to the spatial arrangement of the features. The experiments also show that backward visual masking does produce abrupt termination of the firing of neurons in the temporal cortical visual system, so that the duration of a neuronal response is very short when visual stimuli can just be perceived.
Similar content being viewed by others
References
Baylis GC, Rolls ET, Leonard CM (1985) Selectivity between faces in the responses of a population of neurons in the cortex in the superior temporal sulcus of the monkey. Brain Res 342:91–102
Baylis GC, Rolls ET, Leonard CM (1987) Functional subdivisions of temporal lobe neocortex. J Neurosci 7:330–342
Botzel K, Grusser O-J (1989) Electric brain potentials evoked by pictures of faces and non-faces: a search for “face-specific” EEG-potentials. Exp Brain Res 77:349–360
Bruce C, Desimone R, Gross CG (1981) Visual properties of neurons in a polysensory area in superior temporal sulcus of the macaque. J Neurophys 46:369–384
Desimone R, Albright TD, Gross CG, Bruce C (1984) Stimulusselective properties of inferior temporal neurons in the macaque. J Neurosci 4:2051–2062
Feigenbaum JD, Rolls ET (1991) Allocentric and egocentric spatial information processing in the hippocampal formation of the behaving primate. Psychobiology 19:21–40
Gorea A, Julesz B (1990) Context superiority in a detection task with line-element stimuli: a low-level effect. Perception 19:5–16
Hasselmo ME, Rolls ET, Baylis GC (1989a) The role of expression and identity in the face-selective responses of neurons in the temporal visual cortex of the monkey. Behav Brain Res 32:203–218
Hasselmo ME, Rolls ET, Baylis GC, Nalwa V (1989b) Object-centered encoding by face-selective neurons in the cortex in the superior temporal sulcus of the monkey. Exp Brain Res 75:417–429
Humphreys GW, Bruce V (1989) Visual cognition. Erlbaum, London
Jeffreys DA (1989) A face-responsive potential recorded from the human scalp. Exp Brain Res 78:193–202
Jeffreys DA, Tukmachi ESA (1992) The vertex-positive scalp potential evoked by faces and by objects. Exp Brain Res 91:340–350
Merrill EG, Ainsworth A (1972) Glass-coated platinum-plated tungsten microelectrodes. Med Biol Eng 10:662–672
Oram MW, Perrett DI (1992) Time course of neural responses discriminating different views of the face and head. J Neurophysiol 68:70–84
Perrett DI, Rolls ET, Caan W (1982) Visual neurons responsive to faces in the monkey temporal cortex. Exp Brain Res 47:329–342
Perrett DI, Mistlin AJ, Chitty AJ, Smith PAJ, Potter DD, Broenniman R, Harries M (1988) Specialized face processing and hemisphere asymmetry in man and monkey: evidence from single unit and reaction time studies. Behav Brain Res 29:245–258
Purcell DG, Stewart AL (1986) The face detection effect. Bull Psychonomic Soc 24:118–120
Purcell DG, Stewart AL (1988) The face-detection effect: configuration enhances detection. Percept Psychophys. 43:355–366
Rolls ET (1984) Neurons in the cortex of the temporal lobe and in the amygdala of the monkey with responses selective for faces. Hum Neurobiol 3:209–222
Rolls ET (1991) Neural organisation of higher visual functions. Curr Opin Neurobiol 1:274–278
Rolls ET (1992) Neurophysiological mechanisms underlying face processing within and beyond the temporal cortical visual areas. Philos Trans R Soc Lond Biol 335:11–21
Rolls ET (1994a) Brain mechanisms for invariant visual recognition and learning. Behav Proc (in press)
Rolls ET (1994b) Learning mechanisms in the temporal lobe visual cortex. Behav Brain Res (in press)
Rolls ET, Burton MJ, Mora F (1976) Hypothalamic neuronal responses associated with the sight of food. Brain Res 111:53–66
Rolls ET, Sanghera MK, Roper-Hall A (1979) The latency of activation of neurons in the lateral hypothalamus and substantia innominata during feeding in the monkey. Brain Res 164:121–135
Rolls ET, Tovee MJ (1994) Processing speed in the cerebral cortex and the neurophysiology of visual masking. Proc R Soc B 257:9–15
Rolls ET, Yaxley S, Sienkiewicz ZJ (1990) Gustatory responses of single neurons in the orbitofrontal cortex of the macaque monkey. J Neurophysiol 64:1055–1066
Schiller PH (1968) Single unit analysis of backward visual masking and metacontrast in cat lateral geniculate nucleus. Vision Res 8:855–866
Thorpe SJ, Imbert M (1989) Biological constraints on connectionist models. In: Pfeifer R, Schreter Z, Fogelman-Soulie F (eds) Connectionism in perspective. Elsevier, Amsterdam, pp 63–92
Tovee MJ, Cohen-Tovee EM (1993) The neural substrates of face processing models: a review. Cogn Neuropsychol 10:505–528
Tovee MJ, Rolls ET, Treves A, Bellis RP (1993) Information encoding and the responses of single neurons in the primate temporal visual cortex. J Neurophysiol 70:640–654
Treves A, Rolls ET, Tovee MJ (1995) On the time required for recurrent processing in the brain. Proc Natl Acad Sci USA
Valentine T (1991) A unified account of the effect of distinctiveness, inversion, and race in face recognition. Q J Exp Psychol 43A:161–204
Valentine T, Bruce V (1986) The effects of distinctiveness in recognizing and classifying faces. Perception 15:525–535
Yamane S, Kaji S, Kwano K (1988) What facial features activate face neurons in the inferotemporal cortex of the monkey? Exp Brain Res 73:209–214
Young MP, Yamane S (1992) Sparse population coding of faces in the inferotemporal cortex. Science 256:1327–1331
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rolls, E.T., Tovee, M.J., Purcell, D.G. et al. The responses of neurons in the temporal cortex of primates, and face identification and detection. Exp Brain Res 101, 473–484 (1994). https://doi.org/10.1007/BF00227340
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00227340