Abstract
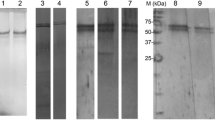
Type-1 fimbriated Salmonella typhimurium was found to adhere to rat intestinal brush border membrane in a mannose sensitive manner. The maximum binding of the purified fimbriae observed with the rat illeal enterocytes was inhibited by 69.2% in presence of D-mannose. Brush border membrane from rat illeum was isolated, delipidified, solubilised and fractionated by affinity chromatography on type-1 fimbriae coupled Sepharose CL 4B column. Sodium dodecyl sulphate polyacrylamide gel electrophoresis of the material eluted from the column with D-mannose revealed a single band of molecular weight 60 kDa. The direct binding of this affinity eluted glycoprotein to the purified type-1 fimbriae was demonstrated by a modified Western blot experiment. Our findings suggest that the 60 kDa glycoprotein may serve as a receptor for the type-1 fimbriae in the rat intestinal brush border membrane and thereby may help in mediating bacterial adherence to the host epithelium.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Harries JT: In: Acute diarrhoea in childhood. Ciba foundation Symposium 42, Elsevier, New York, 1976, pp 3–16
Tzipori S (ed): Infectious Diarrhoea in the young. Strategies for control in humans and animals. Elsevier Sci Publ B V (Biochemical Division), Amsterdam, 1985
World Health Organization programme for control of diarrhoea diseases — Interim Programme Report WHO/CDD 84: 10, 1983
Dugoid JP, Darekar MR, Wheater DWF: Fimbriae and infectivity in Salmonella typhimurium. J Med Microbiol 9: 459–473, 1976
Meadow L, Schneider H, Beem MO: Salmonella enteritidis bacterimia in childhood. J Infect Dis 152: 185–189, 1985
Elsinghorst EA, Banan LS, Kopeeko DJ: Penetration of human intestinal epithelial cells by Salmonella: molecular cloning and expression of Salmonella typhi invasion determinants in Escherichia coli. Proc Natl Acad Sci (USA) 86: 5173–5177, 1989
Galan JE, Curtiss R: Cloning and molecular characterization of genes whose products allow Salmonella typhimurium to penetrate tissue culture cells. Proc Natl Acad Sci (USA) 86: 6383–6387, 1989
Tavendale A, Jardine CKH, Old DC, Dugoid JP: Hemagglutinins and adhesion of Salmonella typhimurium to HEP-2 and Hela cells. J Gen Microbiol 16: 371–380, 1983
Lindquist BL, Lebenthal E, Lee PC, Stinson MW, Merrick FM: Adherence of Salmonella typhimurium to small intestinal enterocytes of the rat. Infect Immun 55: 3044–3050, 1987
Ghosh S, Mittal A, Ganguly NK: Purification and characterization of distinct type of mannose sensitive fimbriae from Salmonella typhimurium. FEMS Microbiol Lett 115: 229–234, 1994
Basu S, Sarkar M, Mandal C: A single step purification of a sialic acid binding lectin (Achatinin) from Achatina fulica snail. Mol Cell Biochem 71: 149–157, 1986
Engvall E, Perlmann P: Enzyme linked immunosorbent assay. ELISA. III. Quantitation of specific antibodies by enzyme labelled anti-immunoglobulin in antigen-coated tubes. J Immunol 109: 129–135, 1972
Moch T, Hoschutzky, H, Hacker J, Kroncke KD and Jann K: Isolation and characterization of the α-sialyl-β-2,3-galactosyl-specific adhesin from fimbriated Escherichia coli. Proc Natl Acad Sci USA, 84: 3462–3466, 1987
Kessler M, Acuto O, Storelli C, Murer H, Semenza H: A modified procedure for the rapid preparation of efficiently translocating vesicles from small intestinal brush border membranes. Their use in investigating some properties of D-glucose and choline transport systems. Biochem Biophys Acta 506: 136–154, 1978
Pinkus LM: Separation and use of enterocytes. Method enzymol 77: 154–162, 1981
Laemmli UK: Clevage of structural proteins during the assembly of the head of bacteriophage T4. Nature (London) 227: 680–685, 1970
March SC, Parikh I, Cuatrecasas P: A simplifies method for cyanogen bromide activation of Agarose for affinity chromatography. Anal Biochem 60: 149–152, 1973
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ: Protein measurement with Folin phenol reagent. J Biol Chem 193: 265–275, 1951
Towbin H: Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets. Procedure and some application. Proc Natl Acad Sci (USA) 76: 4350–4354, 1979
Bradford MM: A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein dye binding. Anal Biochem. 72: 248–254, 1976
Firon N, Ashkenazi S, Mirelman D, Ofeck I, Sharon N: Aromatic aglycosides of mannose are powerful inhibitors of the adherence of type-1 frmbriated E. coli to yeast and intestinal epithelial cells. Infect Immun 55: 472–476, 1987
Sharon N, Lis H: Glycoproteins. In: H. Neurath, R.L. Hill (eds). The proteins, 3rd edn, Vol. 5 Academic, New York 1982, pp 1–144
Rodriguez-Ortega M, Ofeck I, Sharon N: Membrane glycoproteins of human polymorphonuclear leukocytes that act as receptors for mannose specific E. coli infect Immun 55: 968–973, 1987
Laux DC, McSweegan EF, Williams TJ, Wadolkowski EA, Cohen PS: Identification and characterization of mouse small intestine mucosal receptors for E. coli K-12 (k 88ab) Infect Immun 52(1): 18–25, 1986
Sellwood R: An intestinal receptor for the k88 antigen of porcine enteroxigenic E. coli. In: E.C. Boedecker (ed). Attachment of organisms to the gut mucosa; CRC Press, Inc Boca Raton, Fla 10(2) 167–175, 1984
Firon N, Ofek I, Sharon N: Interaction of mannose containing oligosaccharides with the fimbrial lectin E. coli. FEMS Microbiol Lett 7: 265–269, 1982
Anderson MJ, Whitehead JS, Kim TS: Interaction of E. coli k88 antigen with porcine intestinal brush border membranes. Infect Immun 29: 897–901, 1980
Gibbons RA, Jones GW, Sellwood R: An attempt to identify the intestinal receptor for the k88 adhesin by means of a hemagglutination inhibition test using glycoprotein and fraction from sow colostrum. J Gen Microbiol 86: 228–240, 1975
Patricis L, Welin CA, Cohen PS: Presence of k88 — specific receptors in procine illeal mucus is age dependent. Infect Immun 58(10): 3178–3182, 1990
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ghosh, S., Mittal, A., Vohra, H. et al. Interaction of a rat intestinal brush border membrane glycoprotein with type-1 fimbriae of Salmonella typhimurium . Mol Cell Biochem 158, 125–131 (1996). https://doi.org/10.1007/BF00225838
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00225838