Summary
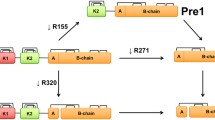
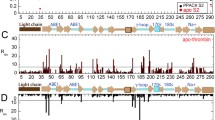
Human pro-coagulant α-thrombin may be proteolyzed under controlled conditions to the non-coagulant β- and γ-thrombin forms. These derivative forms nonetheless retain esterase and amidase activities with small substrates as well as several other thrombin functions. Structurally, human γ-thrombin consists of three non-covalently associated fragments which retain structural integrity as measured by several spectroscopic criteria as well as enzymatic function. The protein folding characteristics of three-chain γ-thrombin indicate that each fragment (domain) contains sufficient information to result in a correct renaturation of protein conformation. Those subtle structural differences which distinguish γ- from α-thrombin are most likely the obstructions to fibrinogen binding which account for the loss of clotting activity.
Similar content being viewed by others
References
Fenton, J. W. II, 1981. Ann. N.Y. Acad. Sci. 370: 468–495.
Fenton, J. W. II, Landis, B. H., Walz, D. A. and Finlayson, J. S., 1977. In: Chemistry and Biology of Thrombin (Lundblad, R. L., Fenton, J. W. 11 and Mann, K. G., eds.), pp. 43–70, Ann Arbor Science Publishers, Michigan.
Lorand, L., 1975. In: Proteases and Biological Control (Reich, E., Rifkin, D. B. and Shaw, E., eds.), pp. 79–84, Cold Spring Harbor Publ., N.Y.
Lollar, P., Hoak, J. C. and Owen, W. G., 1980. J. Biol. Chem. 255:10279–10283.
Galdal, K. S., Evensen, S. A. and Brosstad, F., 1982. Thrombosis Res. 27: 575–584.
Goldsmith, J. C. and Kisker, C. T., 1982. Thrombosis Res. 25:131–136.
Janssens, W. J. and Verhaeghe, R. H., 1982. Blood Vessels 19: 126–134.
Bar-shavit, R., Kahn, A., Fenton, J. W. II and Wilner, G. D., 1983. J. Cell. Biol. 96: 282–285.
Fenton, J. W. 11, Landis, B. H., Walz, D. A., Bing, D. H., Feinman, R. D., Zabinski, M. P., Sonder, S. A., Berliner, L. J. and Finlayson, J. S., 1979. In: The Chemistry and Physiology of the Human Plasma Proteins (Bing, D. H., ed.), pp. 151–183, Pergamon Press, N.Y.
Hageman, T. C., Endres, G. F. and Scheraga, H. A., 1975. Arch. Biochem. Biophys. 171: 327–336.
Wilner, G. D., Danitz, M. P., Fenton, J. W. II and Mudd, M. S., 1979. Clin. Res. 27: 642A.
Chang, T.-L., Feinman, R. D., Landis, B. H. and Fenton, J. W. II, 1979. Biochemistry 18: 113–119.
Bauer, R. S., Chang, T.-L. and Berliner, L. J., 1980. J. Biol. Chem. 255: 5900–5903.
Chang, T.-L., Bauer, R. S. and Berliner, L. J., 1980. J. Biol. Chem. 255: 5904–5906.
Landis, B. H., Koehler, K. A. and Fenton, J. W. II, 1981. J. Biol. Chem. 256: 4604–4610.
Villanueva, G. B., 1981. Biochemistry 20: 6519–6525.
Berliner, L. J. and Shen, Y. Y. L., 1977. In: Chemistry and Biology of Thrombin(Lundblad, R. L., Fenton, J. W. II and Mann, K. G., eds.), pp. 197–216, Ann Arbor Science Publishers, Michigan.
Berliner, L. J., Bauer, R. S., Chang, T.-L., Shen, Y. Y. L. and Fenton, J. W. II, 1981. Biochemistry 20: 1831–1837.
Conery, B. G. and Berliner, L. J., 1983. Biochemistry 22: 369–375.
Berliner, L. J. and Shen, Y. Y. L., 1977. Biochemistry 16: 4622–4626.
Berliner, L. J. and Shen, Y. Y. L., 1977. Thrombosis Res. 12: 15–25.
Koehler, K. A. and Magnusson, S., 1974. Arch. Biochem. Biophys. 160: 175–184.
Downing, M. R., Butkowski, R. J., Clark, M. M. and Mann, K. G., 1975. J. Biol. Chem. 250: 8897–8906.
Lundblad, R. L., Noyes, C. M., Mann, K. G. and Kingdon, H. S., 1979. J. Biol. Chem. 254: 8524–8528.
Lundblad, R. L., Kingdon, H. S. and Mann, K. G., 1976. Meth. Enzymol. 45: 156–176.
Lottenberg, R., Hall, J. A., Fenton, J. W. II and Jackson, C. M., 1982. Thrombosis Res. 28: 313–332.
Tam, S. W., Fenton, J. W. II and Detwiler, T. C., 1980. J. Biol. Chem. 255: 6626–6632.
Charo, I. F., Feinman, R. D. and Detwiler, T. C., 1977. J. Clin. Invest. 60: 866–873.
Snider, R. M. and Richelson, E., 1983. Science 221: 566–568.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Berliner, L.J. Structure-function relationships in human α- and γ-thrombins. Mol Cell Biochem 61, 159–172 (1984). https://doi.org/10.1007/BF00222493
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00222493