Summary
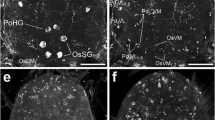
Salivary of the ixodid tick Boophilus microplus Canestrini are at least partially innervated by a branch of the pedipalpal nerve. Axons containing both large granular and smaller agranular vesicles were observed within the acini associated with all types of secretory cells. A modification of the Falk-Hillarp histochemical technique was used to demonstrate discrete areas of fluorescence within the salivary acini. It is suggested that the transmitter involved with the control of salivary activities is a catecholamine and may even be dopamine.
Similar content being viewed by others
References
Berridge, M.J., Patel, N.G.: Insect salivary glands: stimulation of fluid secretion by 5-hydroxytryptamine and adenosine-3′,5′-monophosphate. Science 162, 462–463 (1968)
Binnington, K.C., Tatchell, R.J.: The nervous system and neurosecretory cells of Boophilus microplus (Acarina, Ixodidae). Z. wiss. Zool. Leipzig 185, 193–206 (1973)
Bland, K.P., House, C.R. Ginsborg, B.L., Lazlo, I.: Catecholamine transmitter for salivary secretion in the cockroach. Nature (Lond.) New Biol. 244, 26–27 (1973)
Bowser-Riley, F., House, C.R.: The actions of some putative neurotransmitters on the cockroach salivary gland. J. exp. Biol. 64, 665–676 (1976)
Burgess, L., Rempel, J.G.: The stomadeal nervous system, the neurosecretory system and the gland complex in Aedes aegypti (L) Diptera: Culicidae). Canad. J. Zool. 44, 731–765 (1966)
Coons, L.B., Roshdy, M.A.: Fine structure of the salivary glands of unfed Dermacentor variabilis (SAY) (Ixoidea, Ixodidae). J. Parasit. 59, 900–912 (1973)
Coons, L.B., Roshdy, M.A., Axtell, R.C.: Fine structure of the central nervous system of Dermacentor variabilis (SAY), Amblyomma americanum (L.), and Argus arboreus Kaiser, Hoogstraal, and Kohls (Ixodoidea). J. Parasit. 60, 687–698 (1974)
Corrodi, H., Jonsson, G.: The formaldehyde fluorescence method for the histochemical demonstration of biogenic amines. A review of the methodology. J. Histochem. Cytochem. 15 (2) 65–78 (1967)
Falk, B., Hillarp, N.A., Thieme, G., Torp, A.: Fluorescence of catecholamines and related compunds condensed with formaldehyde. J. Histochem. Cytochem. 10, 348–354 (1962)
Fry, J.P., House, C.R., Sharman, D.F.: An analysis of the catecholamine content of the salivary gland of the cockroach. Brit. J. Pharmacol. 51, 116P-117P (1974)
Grillo, M.A.: Electron microscopy of sympathetic tissues. Pharmacol. Rev. 18, 387–399 (1966)
Håkanson, T., Sundler, F.: Formaldehyde condensation at reduced temperature. Increased sensitivity and specificity of the fluorescence method for demonstrating primary catecholamines. J. Histochem. Cytochem. 22, 887–894 (174)
Kaufman, W.R.: The influence of various factors on fluid secretion by in vitro salivary glands of ixodid ticks. J. exp. Biol. 64, 727–742 (2976)
Kaufman, W.R., Phillips, J.E.: Ion and water balance in the ixodid tick, Dermacentor andersoni. II. Mechanism and control of salivary secretion. J. exp. Biol. 58, 537–547 (1973)
Klemm, N.: Monoamine containing nervous fibres in foregut and salivary gland of the desert locust, Schistocerca gregaria Forskal (Orthoptera-Acridae). Comp. Biochem. Physiol. 43A, 207–211 (1972)
Lane, N.J.: Fine structure of a lepidopteran nervous system and its accessibility to peroxidase and lanthanum. Z. Zellforsch. 131, 205–222 (1972)
Megaw, M.W.J.: Studies on the water balance mechanism of the tick Boophilus microplus Canestrini. Comp. Biochem. Physiol. 48A, 115–125 (1974)
Megaw, M.W.J.: Structure and function of the salivary glands of the tick, Boophilus microplus. Ph.D. Thesis, University of Cambridge (1976)
Megaw, M.W.J., Robertson, H.A.: Dopamine and noradrenaline in the salivary glands and brain of the tick Boophilus microplus: Effect of reserpine. Experientia Basel 30, 1261–1262 (1974)
Meridith, J., Kaufman, W.L.: A proposed site of fluid secretion in the salivary gland of the ixodid tick, Dermacentor andersoni. Parasitology 67, 205–217 (1973)
Obenchain, F.D., Oliver, J.H.: Peripheral nervous system of the ticks, Amblyomma tuberculatum Marx and Argus radiatus Railliet (Acari: Ixodoidea). J. Parasitol. 62, 811–817 (1976)
Robertson, H.A.: The innervation of the salivary gland of the moth, Manduca sexta. Cell Tiss. Res. 148, 237–245 (1974)
Robertson, H.A.: The innervation of the salivary gland of the moth Manduca sexta: evidence that dopamine is the transmitter. J. exp. Biol. 63, 413–419 (1975)
Ruskell, G.L.: The fine structure of nerve terminations in the lacrimal gland of monkeys. J. Anat. (Lond.) 103, 65–76 (1968)
Sauer, J.R., Frick, J.H., Hair, J.A.: Control of 36CL uptake by isolated salivary glands of the lone star tick. J. Insect. Physiol. 20, 1771–1778 (1974)
Tandler, B., Ross, L.L.: Observations of nerve terminals in human labial salivary glands. J. Cell Biol. 42, 339–343 (1969)
Venable, J.H., Coggeshall, R.: A simplified lead citrate stain for use in electron microscopy. J. Cell Biol. 24, 407–408 (1965)
Watari, N.: Fine structure of nervous elements in the pancreas of some vertebrates. Z. Zellforsch. 85, 291–314 (1968)
Whitehead, A.T.: The innervation of the salivary gland of Periplaneta americana (L.): light and electron microsopic observations. J. Morph. 135, 483–506 (1971)
Author information
Authors and Affiliations
Additional information
This work formed part of a thesis submitted to the University of Cambridge for the degree of Ph.D. and was supported by a studentship from the Science Research Council. I would like to thank Drs. D.J. Beadle, M.J. Berridge and H.A. Robertson for their encouragement and advice
Rights and permissions
About this article
Cite this article
Megaw, M.W.J. The innervation of the salivary gland of the tick, Boophilus microplus . Cell Tissue Res. 184, 551–558 (1977). https://doi.org/10.1007/BF00220978
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00220978