Summary
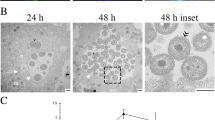
When amoebae are injected with heterologous cytoplasm from another type of amoeba, cellular organelles are segregated and degenerate within membrane-bounded spaces in the host's cytoplasm. In the present study, the role of the nucleus in this response was tested by injecting heterologous cytoplasm from Pelomyxa carolinensis into enucleate and nucleate Amoeba discoides. Samples were prepared for ultrastructural study at intervals between 15 min and 2d after the operation. In enucleate recipients of heterologous cytoplasm, organelles and cellular debris were observed in vacuole-like spaces indistinguishable in morphology and frequency from those of nucleated recipients. When donor cytoplasm included thoria particles, the electron-dense tracer was found along with degenerating organelles in the vacuole-like structures, confirming the presence there of injected cytoplasm. Changes of comparable magnitude were not observed when either nucleated or enucleated cells were injected with homologous cytoplasm. The results indicate that nuclear activity is not necessary for the ultrastructurally observable responses of amoebae to heterologous cytoplasm. It is suggested therefore that the cytoplasm possesses a mechanism for recognition of at least some types of heterologous organelles.
Similar content being viewed by others
References
Cameron JM, Hawkins SE (1976) Inhibition of cell division in amoebae: the incorporation of tritiated precursors into Amoeba proteus after the injection of non-homologous cytoplasm. J Cell Sci 20:525–537
Chalkley HW (1930) Stock cultures of ameba. Science 71:442
Daniels EW (1962) Limits of transplantation tolerance in large amoebae. I. Microfusion studies using Amoeba proteus, Pelomyxa illinoisensis and three strains of Pelomyxa carolinensis. J. Protozool 9:183–187
Daniels EW (1973) Ultrastructure. In: Jeon KW (ed) The biology of amoeba. Academic Press, New York, pp 125–169
Flickinger CJ (1968) The effects of enucleation on the cytoplasmic membranes of Amoeba proteus. J. Cell Biol 37:300–315
Flickinger CJ (1969) The development of Golgi complexes and their dependence upon the nucleus in amebae. J Cell Biol 43:250–262
Flickinger CJ (1973) Cellular membranes of amoeba. In: Jeon KW (ed) The biology of amoeba. Academic Press, New York, pp 171–199
Flickinger CJ (1974a) Radioactive labeling of the Golgi apparatus by micro-injection of individual amebae. Exp Cell Res 88:415–418
Flickinger CJ (1974) The fine structure of four “species” of Amoeba. J Protozool 21:59–68
Flickinger CJ, Read GA, Kabana EM (1980) Structural responses of amoebae to injection of heterologous cytoplasm. J Cell Sci in press
Jeon KW (1970) Micromanipulation of amoeba nuclei. Methods Cell Physiol 4:179–194
Jeon KW (1972) Development of cellular dependence on infective-organisms: Micrurgical studies in amoebas. Science 176:1122–1123
Jeon KW, Danielli JF (1971) Micrurgical studies with large free-living amebas. Int Rev Cytol 30:49–89
Jeon KW, Jeon MS (1976) Endosymbiosis in amoebae: recently established endosymbionts have become required cytoplasmic components. J Cell Physiol 89:337–344
Karnovsky MJ (1965) A formaldehyde-glutaraldehyde fixative of high osmolality for use in electron microscopy. J Cell Biol. 27:137A
Ord MJ (1968) The viability of the anucleate cytoplasm of Amoeba proteus. J Cell Sci 3:81–88
Prescott DM, Carrier RF (1964) Experimental procedures and cultural methods for Euplotes eurystomus and Ameba proteus. Methods Cell Physiol 1:85–95
Author information
Authors and Affiliations
Additional information
Supported by grants from the American Cancer Society (VC-169A, B, C; CD-58D)
The authors are indebted for technical assistance to Mrs. Christine Folsom-Kovarik, Ms. Kathleen Coogan, and Ms. Ellen Kabana
Rights and permissions
About this article
Cite this article
Flickinger, C.J., Read, G.A. Capacity of enucleated amoebae to recognize and respond to heterologous cytoplasm. Cell Tissue Res. 210, 395–401 (1980). https://doi.org/10.1007/BF00220197
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00220197