Summary
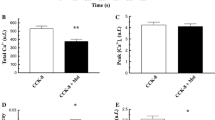
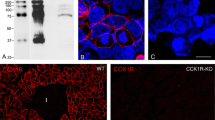
Cholecystokinin (CCK) and acetylcholine, at concentrations greater than those required for maximal pancreatic enzyme secretion, elicit a submaximal secretory response. The mechanism for this “secretagogue-induced unresponsiveness” is unknown. Using isolated pancreatic acini of the mouse, we now find that high concentrations of secretagogues also induce a profound alteration in acinar morphology, characterized by the formation of spherical protrusions on the basal surface of the cells. Since both the determination of cell shape and exocytosis may involve calcium and contractile proteins, we used a calcium-free medium and cytochalasin B (CB) to evaluate the importance of a contractile mechanism in the secretory and morphological effects of high concentrations of CCK-octapeptide (CCK8). Incubation in a calcium-free medium partially blocked CCK-induced unresponsiveness, but brought about dissociation of the acini. CB at a concentration of 3 μg/ml caused the disappearance of apical microfilaments and, most strikingly, completely prevented the morphological alteration induced by CCK8. Furthermore, CB converted the biphasic dose-response curve for CCK8-induced amylase release to a monophasic shape, such that the amylase release stimulated by a high concentration of CCK8 (10 nM) was augmented. It is concluded, therefore, that a contractile process involving microfilaments may mediate “secretagogue-induced unresponsiveness” in pancreatic acinar cells.
Similar content being viewed by others
References
Abdelmoumene S, Gardner JD (1980) Cholecystokinin-induced desensitization of enzyme secretion in dispersed acini from guinea pig pancreas. Am J Physiol 239:G272-G279
Adelstein RS, Scordilis SP, Trotter JA (1979) The cytoskeleton and cell movement: general considerations. Methods Achiev Exp Pathol 8:1–41
Amsterdam A, Jamieson JD (1974) Studies on dispersed pancreatic exocrine cells, I. Dissociation technique and morphologic characteristic of separated cells. J Cell Biol 63:1037–1056
Bartfai T (1979) Preparation of metal-chelate complexes and the design of steady-state kinetic experiments involving metal nucleotide complexes. Adv Cyclic Nucleotide Res 10:219–242
Bauduin H, Stock C, Vincent D, Grenier JF (1975) Microfilamentous system and secretion of enzyme in the exocrine pancreas, effect of cytochalasinB. J Cell Biol 66:165–181
Brown SS, Spudich JA (1981) Mechanism of action of cytochalasin. Evidence that it binds to actin filament ends. J Cell Biol 88:487–491
Butcher FR (1978) Regulation of exocytosis. In: Litwack G (ed) Biochemical actions of hormones; Vol 5. Academic Press, New York, pp 54–99
Chandler DE, Williams JA (1978) Intracellular divalent cation release in pancreatic acinar cells during stimulus-secretion coupling. I. Use of chlorotetracycline as a fluorescent probe. J Cell Biol 76:371–385
Dedman JR, Brinkley BR, Means AR (1979) Regulation of microfilaments and microtubules by calcium and cyclic AMP. Adv Cyclic Nucleotide Res 11:131–174
Folsch UR, Wormsley KG (1973) Pancreatic enzyme response to secretin and cholecystokininpancreozymin in the rat. J Physiol (Lond) 102:115–125
Gardner JD, Costenbader CL, Uhlemann ER (1979) Effect of extracellular calcium on amylase release from dispersed pancreatic acini. Am J Physiol 236:E754-E762
Godman GC, Miranda AF (1978) Cellular contractility and the visible effects of cytochalasin. In: Tanenbaum SW (ed) Frontiers of biology 46. North Holland, Amsterdam, pp 277–430
Jung DH (1980) Preparation and application of Procion Yellow starch for amylase assay. Clin Chim Acta 100:7–11
Korc MJ, Williams JA, Goldfine ID (1979) Stimulation of the glucose transport system in mouse pancreatic acini by cholecystokinin and analogues. J Biol Chem 254:7624–7629
Meldolesi J, Castiglioni G, Parma R, Nassivera N, DeCamilli P (1978) Calcium dependent disassembly and reassembly of occluding junctions in guinea pig pancreatic acinar cells. Effects of drugs. J Cell Biol 79:156–172
Savion N, Selinger Z (1978) Morphological changes in rat pancreatic slices associated with inhibition of enzyme secretion by high concentrations of secretagogues. J Cell Biol 76:467–482
Williams JA (1975) An in vitro evaluation of possible cholinergic and adrenergic receptors affecting pancreatic amylase secretion. Proc Soc Exp Biol Med 150:513–516
Williams JA (1977) Effects of cytochalasin B on pancreatic acinar cell structure and secretion. Cell Tissue Res 179:453–466
Williams JA, Korc M, Dormer RL (1978) Action of secretagogues on a new preparation of functionally intact, isolated pancreatic acini. Am J Physiol 235:E517-E524
Williams JA, Sankaran H, Korc M, Goldfine ID (1981) Receptors for cholecystokinin and insulin in isolated pancreatic acini: hormonal control of secretion and metabolism. Fed Proc 40:2497–2502
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Burnham, D.B., Williams, J.A. Effects of high concentrations of secretagogues on the morphology and secretory activity of the pancreas: A role for microfilaments. Cell Tissue Res. 222, 201–212 (1982). https://doi.org/10.1007/BF00218300
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00218300