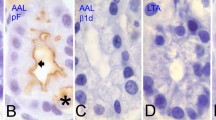
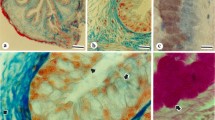
Summary
The cellular localization of an aminopeptidase N homologous to the brush-border intestinal enzyme and that of human blood group A-substances were investigated using the immunofluorescence technique on thin frozen sections (200 nm) of the digestive tract and associated glands of A+ and A− rabbits. Aminopeptidase N was found to be a common specific marker of both the apical region of plasma membrane of acinar cells in submaxillary and parotid glands and pancreas and the brush border of jejunum and colon absorbing cells. In hepatocytes, the enzyme was localized in the sinusoidal domains. Soluble A-substances were present in mucus secretory granules of intestinal goblet cells and those of stomach and gall bladder mucous cells. In contrast, the mucous acini of sublingual and submaxillary glands were devoid of A-antigenicity. The columnar cells of striated ducts of these glands exhibited A-antigenicity. Soluble A-substances were also found in zymogen granules of parotid and pancreas acinar cells and those of stomach chief cells. Moreover, in all cells secreting A-substances, and in the non-secreting absorbing intestinal cells, the glycoproteins of the plasma membrane bore A-determinants. Aminopeptidase N was one of the membrane-bound glycoproteins that bore A-determinants in cells that expressed A-antigenicity.
Similar content being viewed by others
References
Baig MM, Aminoff D (1972) Glycoproteins and blood group activity. J Biol Chem 247:6111–6118
Bernadac A, Gorvel JP, Feracci H, Maroux S (1984) Human blood group A-like determinants as marker of the intracellular pools of glycoproteins in secretory and absorbing cells in A+ rabbit jejunum. Biol Cell 50:31–36
Blobel G, Dobberstein B (1975) Reconstitution of functional rough microsomes from heterologous components. J Cell Biol 67:835–851
Burnette NN (1981) “Western blotting”: electrophoretic transfer of proteins from sodium dodecyl sulfate-polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Analyt Biochem 112:195–203
Coudrier E, Reggio H, Louvard D (1983) Characterization of an integral membrane glycoprotein associated with the microfilaments of pig intestinal microvilli. EMBO J 2:469–475
Farquhar MG, Palade GE (1981) The Golgi apparatus (complex)-(1954–1981)-from artefact to center stage. J Cell Biol 9:775–1035
Feracci H, Maroux S (1980) Rabbit intestinal aminopeptidase N. Purification and molecular properties. Biochim Biophys Acta 599:448–463
Feracci H, Bernadac A, Hovsépian S, Fayet G, Maroux S (1981) Aminopeptidase N is a marker for the apical pole of porcine thyroid epithelial cells in vivo and in culture. Cell Tissue Res 221:137–146
Feracci H, Bernadac A, Gorvel JP, Maroux S (1982) Localization by immunofluorescence and histochemical labeling of aminopeptidase N in relation to its biosynthesis in rabbit and pig enterocytes. Gastroenterology 82:317–324
Fukasawa KM, Fukasawa K, Sahara N, Harada M, Kondo Y, Nagatsu I (1981) Immunohistochemical localisation of dipeptidyl aminopeptidase IV in rat kidney, liver and salivary glands. J Histochem Cytochem 20:337–343
Gorvel JP, Wisner-Provost A, Maroux S (1982) Identification of glycoprotein bearing human blood group A determinants in rabbit enterocyte plasma membranes. FEBS Lett 143:17–20
Gossrau R (1981) Investigation of proteinases in the digestive tract using 4-methoxy-2-naphthylamine (MNA) substrates. J Histochem Cytochem 29–3A:464–480
Harboe N, Ingild A (1973) Immunization, isolation of immunoglobulins, estimation of antibody titre. In: Axelsen NY, Kroll J, Weeke B (eds) A manual of quantitative immunoelectrophoresis. Methods and applications. Universitetsforlated, Oslo, pp 161–164
Hinglais N, Bretton R, Rouchon M, Oriol R, Bariety J (1981) Ultrastructural localization of blood group A antigen in normal human kidneys. J Ultrastruct Res 74:34–35
Hütter HJ, Heider A, Neef L, Haschen RJ (1982) Alaninaminopeptidase menschlicher Plazenta — Reinigung und Eigenschaften. Z Med Labor Diagn 23:298–305
Ito T, Hiwada K, Kokubu T (1980) Immunological characterization of human membrane bound arylamidases from small intestine, lung, kidney, liver, placenta and renal cell carcinoma. Clin Chim Acta 101:139–143
Kenny JA, Maroux S (1982) Topology of microvillar membrane hydrolases of kidney and intestine. Physiol Rev 62:91–128
Lojda Z (1981) Proteinases in pathology. Usefulness in histochemical methods. J Histochem Cytochem 29–3A:481–493
Louvard D, Maroux S, Desnuelle P (1975a) Topological studies on the hydrolase bound to the intestinal brush border membrane. II. Interactions of free and bound aminopeptidase with a specific antibody. Biochim Biophys Acta 389:389–400
Louvard D, Maroux S, Vannier Ch, Desnuelle P (1975b) Topological studies of the hydrolases bound to the intestinal brush border membrane. I. Solubilization by papain and Triton X 100. Biochim Biophys Acta 375:236–248
Maire I, Dorche C, Rolland MO, Mathieu M, Cotte J (1981) Les formes multiples d'enzymes en biologie clinique. Lyon Pharmaceutique 32:5–9
Maroux S, Feracci H (1982) Structure and biosynthesis of aminopeptidases. In: Fleisher S, Fleisher B (eds) Methods in enzymology. Vol 96, Part J, pp 406–423
Maroux S, Louvard D, Baratti JC (1973) The aminopeptidase from hog intestinal brush border. Biochim Biophys Acta 321:282–295
Moss DW, Butterworth PJ (1974) Isoenzyme tests in diagnosis. In: Moss DW and Butterworth PJ (eds) Enzymology and medicine. Pittman Medical, pp 154–56
Nachlas MM, Crawford DT, Seligman AM (1957) The histochemical demonstration of leucine aminopeptidase. J Histochem Cytochem 5:264–278
Roman L, Hubbard AL (1983) A domain-specific marker for the hepatocyte plasma membrane: localization of leucine aminopeptidase to the bile canalicular domain. J Cell Biol 96:1548–1558
Rouger Ph, Gossens D, Gabe P, Salmon Ch (1981) Distribution of blood group antigens in adult pancreas. Tissue Antigens 18:51–55
Szulman AE (1960) The histological distribution of blood group substances A and B in men. J Exp Med 111:785–800
Tokuyasu KT (1973) A technique for ultracryotomy of cell suspension and tissues. J Cell Biol 57:551–565
Triadou N, Audran E, Roussel M, Zweibaum A, Oriol R (1983) Relationship between secretor status and the expression of ABH blood group antigenic determinants in human intestinal brush border membrane hydrolases. Biochim Biophys Acta 761:231–236
Van Halbeek H, Dorland L, Haverkamp J, Veldink GA, Vliegenthart JFG, Fournet B, Ricart G, Montreuil J, Gathmann WD, Aminoff D (1981) Structure determination of oligosaccharides isolated from A+, H+ and A−, H− hog submaxillary gland mucin glycoproteins by 360-MH2 1H-NMR spectroscopy, permethylation analysis and mass spectrometry. Eur J Biochem 118:487–495
Vannier Ch, Louvard D, Maroux S, Desnuelle P (1976) Structural and topological homology between porcine intestinal and renal brush border aminopeptidase. Biochim Biophys Acta 455:186–199
Wachsmuth ED (1968) Lokalisation von Aminopeptidase in Gewebeschnitten mit einer neuen Immunofluoreszenztechnik. Histochemie 14:282–296
Watkins WM (1972) Blood group substances in glycoproteins. In: Gottschalk A (ed) Biochim Biophys Acta Library vol 5, part B, pp 830–891
Zacharius RM, Zell TE, Morrisson JH, Woodlock JJ (1969) Glycoprotein staining following electrophoresis on acrylamide gels. Anal Biochem 30:148–152
Zweibaum A, Bouhou E (1972) Studies on digestive groups. The A alloantigen-alloantibody system in rabbits. Transplantation 15:291–293
Zweibaum A, Bouhou E (1973) Studies on digestive groups. Tissue distribution of the digestive group A alloantogen in rabbits. Biomedicine 19:544–546
Zweibaum A, Stendler V (1969) Natural isoantibodies specific for tissular group antigens of the colon in dog and rabbit. Nature 223:84
Zweibaum A, Oriol R, Feingold N, Dussaulx E, Rousset M, Chevalier G (1974) Studies on canine secretory alloanligens. Tissue Antigens 4:115–129
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gorvel, JP., Rigal, A., Sarles, J. et al. Aminopeptidase N- and human blood group A-antigenicity along the digestive tract and associated glands in the rabbit. Cell Tissue Res. 239, 241–248 (1985). https://doi.org/10.1007/BF00214925
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00214925