Summary
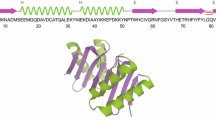
The 1H, 13C and 15N NMR assignments of the backbone and side-chain resonances of rat S100β were made at pH 6.5 and 37°C using heteronuclear multidimensional NMR spectroscopy. Analysis of the NOE correlations, together with amide exchange rate and 1Hα, 13Cα and 13Cβ chemical shift data, provided extensive secondary structural information. Thus, the secondary structure of S100β was determined to comprise four helices (Leu3-Ser18, helix I; Lys29-Leu40, helix II; Gln50-Glu62, helix III; and Phe70-Ala83, helix IV), four loops (Gly19-His25, loop I; Ser41-Glu49, loop II; Asp63-Gly66, loop III; and Cys84-Glu91, loop IV) and two β-strands (Lys26-Lys28, β-strand I and Glu67-Asp69, β-strand II). The β-strands were found to align in an antiparallel manner to form a very small β-sheet. This secondary structure is consistent with predictions that S100β contains two ‘helix-loop-helix’ Ca2+-binding motifs known as EF-hands. The alignment of the β-sheet, which brings the two EF-hand domains of S100β into close proximity, is similar to that of several other Ca2+ ion-binding proteins.
Similar content being viewed by others
References
AbeygunawardanaC., WeberD.J., FrickD.N., BessmanM.J. and MildvanA.S. (1993) Biochemistry, 32, 13071–13080.
AkkeM., DrakenbergT. and ChazinW.J. (1992) Biochemistry, 31, 1011–1020.
AlloreR., O'HanlonD., PriceR., NeilsonK., WillardH.F., CoxD.R., MarksA. and DunnR.J. (1988) Science, 239, 1311–1313.
Amburgey, J.C., Abildgaard, F., Starich, M.R., Shah, S., Hilt, D.C. and Weber, D.J. (1994) Abstracts of the 208th National ACS Meeting, Biological Division, Poster 48, Washington, DC.
AmburgeyJ.C., AbildgaardF., StarichM.R., ShahS., HiltD.C. and WeberD.J. (1995) FASEB J., 9, 1008.
BaudierJ., GlasserN. and GérardD. (1986) J. Biol. Chem., 261, 8192–8203.
BaxA. and DavisD. (1985a) J. Magn. Reson., 65, 355–360.
BaxA. and DavisD. (1985b) J. Magn. Reson., 63, 207–213.
BaxA. and IkuraM. (1991) J. Biomol. NMR, 1, 99–104.
BaxA. and PochapskyS. (1992) J. Magn. Reson., 99, 638–643.
BaxA. and GrzesiekS. (1993) In NMR of Proteins (Eds, CloreG.M. and GronenbornA.M.), CRC Press, Boca Raton, FL, pp. 33–52.
BodenhausenG. and RubenD.J. (1980) Chem. Phys. Lett., 69, 185–188.
BoveyF.A., JelinskiL. and MirauP.A. (1988) Nuclear Magnetic Resonance Spectroscopy, 2nd ed., Academic Press, San Diego, CA.
ChazinW.J., KördelJ., DrakenbergT., ThulinE., BrodinP., GrundströmT. and ForsénS. (1989) Proc. Natl. Acad. Sci. USA, 86, 2195–2198.
DonatoR. (1991) Cell Calcium, 12, 713–726.
DriscollP.C., CloreG.M., MarionD., WingfieldP.T. and GronenbornA.M. (1990) Biochemistry, 29, 3542–3556.
EdisonA.S., AbildgaardF., WestlerW.M., MooberryE.S. and MarkleyJ.L. (1994) Methods Enzymol., 239, 3–79.
Forman-KayJ.D., GronenbornA.M., KayL.E., WingfieldP.T. and CloreM. (1990) Biochemistry, 29, 1566–1572.
GrzesiekS. and BaxA. (1992a) J. Magn. Reson., 96, 432–450.
GrzesiekS. and Bax.A. (1992b) J. Am. Chem. Soc., 114, 6291–6293.
GrzesiekS., AnglisterJ. and BaxA. (1993) J. Magn. Reson. Ser. B, 101, 114–119.
GrzesiekS. and BaxA. (1993) J. Biomol. NMR, 3, 185–204.
HiltD.C. and KligmanD. (1991) In Novel Calcium-Binding Proteins (Ed., HeizmannC.), Springer, Berlin.
IkuraM., BaxA., CloreG.M. and GronenbornA.M. (1990) J. Am. Chem. Soc., 112, 9020–9022.
JeenerJ., MeierB.H., BachmannP. and ErnstR.R. (1979) J. Chem. Phys., 71, 4546–4553.
JiangH., ShahS. and HiltD.C. (1993) J. Biol. Chem., 268, 20502–20511.
KayL.E., MarionD. and BaxA. (1989) J. Magn. Reson., 84, 72–84.
KayL.E., IkuraM., TschudinR. and BaxA. (1990) J. Magn. Reson., 89, 496–514.
KayL.E., KeiferP. and SaarinenT. (1992) J. Am. Chem. Soc., 114, 10663–10665.
KesslerH., GriesingerC., KerssebaumR., WagnerK. and ErnstR.R. (1987) J. Am. Chem. Soc., 109, 607–609.
KligmanD. and MarshakD.R. (1985) Proc. Natl. Acad. Sci. USA, 82, 7136–7139.
KligmanD. and HiltD.C. (1988) Trends Biochem. Sci., 13, 437–443.
KördelJ., ForsénS., DrakenbergT. and ChazinW.J. (1990) Biochemistry, 29, 4400–4409.
KördelJ., SkeltonN.J., AkkeM., PalmerA.G. and ChazinW.J. (1992) Biochemistry, 31, 4856–4866.
KördelJ., SkeltonN.J., AkkeM. and ChazinW.J. (1993) J. Mol. Biol., 231, 711–734.
KretsingerR.H. (1980) CRC Crit. Rev. Biochem., 8, 119–174.
KuwanoR., UsuiH., MaedaT., FukuiT., YamanariN., OhtsukaE., IkeharaM. and TakahashiY. (1984) Nucleic Acids Res., 12, 7455–7465.
LackmannM., RajasekariahP., IismaaS.E., JonesG., CornishC.J., HuS., SimpsonR.J., MoritzR.L. and GeczyC.L. (1993) J. Immunol., 150, 2981–2991.
LiveD.H., DavisD.G., AgostaW.C. and CowburnD. (1984) J. Am. Chem. Soc., 106, 1939–1941.
MaedaT., UsuiH., ArakiK., KuwanoR., TakahashiY. and SuzukiY. (1991) Mol. Brain Res., 10, 193–202.
ManiR.S., BoyesB.E. and KayC.M. (1982) Biochemistry, 21, 2607–2612.
ManiR.S., ShellingJ.G., SykesB.D. and KayC.M. (1983) Biochemistry, 22, 1734–1740.
MarionD., DriscollP.C., KayL.E., WingfieldP.T., BaxA., GronenbornA.M. and CloreG.M. (1989a) Biochemistry, 28, 6150–6156.
MarionD., IkuraM., TschudinR. and BaxA. (1989b) J. Magn. Reson., 85, 393–399.
MooberryE.S., AbildgaardF. and MarkleyJ.L. (1994) Methods Enzymol., 239, 247–256.
MooreB.E. (1965) Biochem. Biophys. Res. Commun., 19, 739–744.
MuhandiramD.R., GuangY.X. and KayL.E. (1993) J. Biomol. NMR, 3, 463–470.
PallaghyP.K., DugganB.M., PenningtonM.W. and NortonR.S. (1993) J. Mol. Biol., 234, 405–420.
PalmerA.G., CavanaghJ., WrightP.E. and RanceM. (1991) J. Magn. Reson., 93, 151–170.
PiantiniU., SørensenO.W. and ErnstR.R. (1982) J. Am. Chem. Soc., 104, 6800–6801.
PowersR., GronenbornA.M., CloreG.M. and BaxA. (1991) J. Magn. Reson., 94, 209–213.
ReddyP., PeterkofskyA. and McKenneyK. (1989) Nucleic Acids Res., 17, 10473–10488.
ReevesR.H., YaoJ., CrowleyM.R., BuckS., ZhangX., YarowskyP., GearhartJ.D. and HiltD.C. (1994) Proc. Natl. Acad. Sci. USA, 91, 5359–5363.
SchleucherJ., SchwendingerM., SattlerM., SchmidtO., SchedletzkyS.J., SørensenO.W. and GriesingerC. (1994) J. Biomol. NMR, 4, 301–306.
ShinnarM., BolingerL. and LeighJ.S. (1989a) Magn. Reson. Med., 12, 88–92.
ShinnarM., EleffS., SubramanianH. and LeighJ.S. (1989b) Magn. Reson. Med., 12, 74–80.
SkeltonN.J., ForsénS. and ChazinW.J. (1990a) Biochemistry, 29, 5752–5761.
SkeltonN.J., KördelJ., ForsénS. and ChazinW.J. (1990b) J. Mol. Biol., 213, 593–598.
SkeltonN.J., KördelJ., AkkeM., ForsénS. and ChazinW.J. (1994) Nature Struct. Biol., 1, 239–245.
SperaS. and BaxA. (1991) J. Am. Chem. Soc., 113, 5490–5492.
StrynadkaN.C.J. and JamesM.N.G. (1989) Annu. Rev. Biochem., 58, 951–998.
SzebenyiD.M.E. and MoffatK. (1986) J. Biol. Chem., 261, 8761–8777.
VanEldikL.J., StaeckerJ.L. and Winningham-MajorF. (1988) J. Biol. Chem., 263, 7830–7837.
VanEldikL.J. and GriffinW.S.T. (1994) Biochim. Biophys. Acta, 1223, 398–403.
WishartD.S., SykesB.D. and RichardsF.M. (1991) J. Mol. Biol., 222, 311–333.
WishartD.S., SykesB.D. and RichardsF.M. (1992) Biochemistry, 31, 1647–1651.
WishartD.S. and SykesB.D. (1994) J. Biomol. NMR, 4, 171–180.
WittekindM. and MuellerL. (1993) J. Magn. Reson. Ser. B, 101, 201–205.
WüthrichK. (1986) NMR of Proteins and Nucleic Acids, Wiley, New York, NY.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Amburgey, J.C., Abildgaard, F., Starich, M.R. et al. 1H, 13C and 15N NMR assignments and solution secondary structure of rat Apo-S100β. J Biomol NMR 6, 171–179 (1995). https://doi.org/10.1007/BF00211781
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00211781