Summary
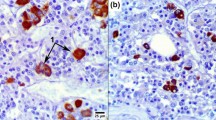
The fine structural characteristics of normal rat corticotrophs stained with anti-porcine ACTH1–39 serum were studied. At the ultrastructure level immunoreactive corticotrophs appear to comprise four distinct cell types: (1) large stellate cells (Siperstein cells) containing granules (170–250 nm in diameter) arranged in a peripheral row and usually embracing an acidophil; (2) elongate spindle-shaped cells (Moriarty cells) in which the secretory granules (170–250 nm in diameter) are distributed in a row or in small clusters in the peripheral cytoplasm; (3) oval or polygonal cells filled only with small secretory granules (130–170 nm in diameter), resembling the “acidophil of small granules type” (Yoshimura et al. 1974); and (4) polygonal or stellate cells filled with secretory granules of varying diameters (180–300 nm in diameter) and occasionally embracing an acidophil. The first type is the most common, but the others are infrequent. It is concluded that the criteria of Siperstein and Miller (1970) do not necessarily include all categories of rat corticotrophs.
Similar content being viewed by others
References
Baker BL, Drummond T (1972) The cellular origin of corticotropin and melanotropin as revealed by immunochemical-staining. Am J Anat 134:385–409
Beauvillain JC, Tramu G (1973) Cellules à activité corticotrope de l'hypophyse du Lérot: superposition des résultats de microscopie optique et de microscopie électronique. C R Acad Sci (Paris) 277 (Série D): 1025–1028
Beauvillain JC, Tramu G, Dubois MP (1975) Characterization by different techniques of adrenocorticotropin and gonadotropin producing cells in lerot pituitary (Eliomys quercinus). A superimposition technique and an immunocytochemical technique. Cell Tissue Res 158:301–317
Bugnon C, Lenys D, Bloch B, Fellman D (1975) Les cellules corticotropes et mélanotropes de l'adénohypophyse chez le chat, le renard, le rat et le foetus humain: étude avec diverse techniques de fluorescence induite, de cyto-immunologie et à l'hématoxyline plombique. C R Soc Biol (Paris) 169:1271–1276
Kumagai T (1980) Human pituitary corticotrophs: Their amphophilic stainability and immunohistochemical characterization. Endocrinol Jpn 27:551–560
Kurosumi K (1968) Functional classification of cell types of the anterior pituitary gland accomplished by electron microscopy. Arch Histol Jpn 29:239–362
Kurosumi K Kobayashi Y (1966) Corticotrophs in the anterior pituitary glands of normal and adrenalectomized rats as revealed by electron microscopy. Endocrinology 78:745–758
Martin R, Weber E, Voigt KH (1979) Localization of corticotropin- and endorphin-related peptide in the intermediate lobe of the rat pituitary. Cell Tissue Res 196:307–319
Millonig G (1962) Further observation on a phosphate buffer for osmium solutions in fixation. Electron microscopy. Fifth International Congress for E.M. Academic Press 2:8
Mollenhauser HH (1964) Plastic embedding mixtures for use in electron microscopy. Stain Tech 39:111–114
Moriarty GC, Halmi NS (1972) Electron microscopic study of the adrenocorticotropin producing cell with the use of unlabeled anti-body and the soluble peroxidase-antiperoxidase complex. J Histochem Cytochem 20:590–603
Moriarty GC, Halmi NS, Moriarty CM (1975) The effects of stress on the cytology and immunocytochemistry of pars intermedia cells in the rat pituitary. Endocrinology 96:1426–1436
Nagata M Mizunaga A, Erna S, Yoshimura F (1980) Various types of the pituitary folliculo-stellate cells involving the Siperstein's corticotroph in the normal rats. Endocrinol Jpn 27:13–22
Nakayama I, Nickerson PA, Skelton FR (1969) An ultrastructural study of the adrenocorticotrophic hormone-secreting cell in the rat adenohypophysis during adrenal cortical regeneration. Lab Invest 21:169–178
Nogami H, Yoshimura F (1980) Prolactin immunoreactivity of acidophils of the small granule type. Cell Tissue Res 211:1–4
Pelletier G, Racadot J (1971) Identification des cellules hypophysaire sécrétant l'ACTH. Z Zellforsch 116:228–239
Phifer PF, Orth DN, Spicer SS (1974) Specific demonstration of the human hypophyseal adrenocorticomelanotrophic (ACTH/MSH) cell. J Clin Endocrin 39:684–692
Roberts JL, Phillips M, Rosa PA, Herbert E (1978) Steps involved in the processing of common precursor forms of adrenocorticotropin and endorphin in cultures of mouse pituitary cells. Biochemistry 17:3609–3618
Siperstein ER, Miller KJ (1970) Further cytophysiologic evidence for the identity of the cells that produce adrenocorticotrophic hormone. Endocrinology 86:451–486
Sternberger LA, Hardy PH, Cuculis JJ, Meyer HG (1970) The unlabeled antibody enzyme method of immunohistochemistry. Preparation and properties of soluble antigen-antibody complex (Horseradish peroxidase-antihorseradish peroxidase) and its use in identification of spirochetes. J Histochem Cytochem 18:315–333
Tramu G, Dubois MP (1977) Comparative cellular localization of corticotropin and melanotropin in lerot adenohypophysis (Eliomys quercinus). An immunohistochemical study. Cell Tissue Res 183:457–469
Yoshimura F, Nogami H (1980) Immunohistochemical characterizations of pituitary stellate cells in rats. Endocrinol Jpn 27:43–51
Yoshimura F, Soji T, Takasaki Y, Kiguchi Y: (1974) Pituitary acidophils with small or medium-sized granules alone in normal and adrenalectomized rats with special reference to possible ACTH secretion. Endocrinol Jpn 21:297–316
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yoshimura, F., Nogami, H. Fine structural criteria for identifying rat corticotrophs. Cell Tissue Res. 219, 221–228 (1981). https://doi.org/10.1007/BF00210144
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00210144