Summary
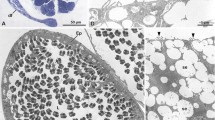
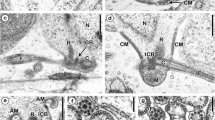
The flagella of the motile sperm cells of Nymphon leptocheles and N. rubrum (Pyonogonida, Arthropoda) exhibit a 12+0 and a 9+0 axoneme pattern, respectively. Central tubules, central sheath, spokes and arms are absent. The doublets are connected by a circular nexus. The functional significance of this axonemal composition is discussed. Aberrant axonemes occurring in high frequencies both within the species and within single specimens are probably explained by the loose axonemal connection, due to the absence of a central complex. This absence is further suggested to have facilitated the evolution from the 9+0 type to the 12+0 type.
Similar content being viewed by others
References
Allen, R. D.: A reinvestigation of cross-sections of cilia. J. Cell Biol. 37, 825–831 (1968)
Baccetti, B., Dallai, R., Fratello, B.: The spermatozoon of Arthropoda. XXII. The 12+0, 14+0 or aflagellate sperm of Protura. J. Cell Sci. 13, 321–335 (1973)
Dallai, R., Bernini, F., Giusti, F.: Interdoublet connection in the sperm flagellar complex of Sciara. J, Submicr. Cytol. 5, 137–145 (1973)
Deurs, B. van: Axonemal 12+0 pattern in the flagellum of the motile spermatozoon of Nymphon leptocheles. J. Ultrastruct. Res. 42, 594–598 (1973)
Deurs, B. van: Spermatology of some Pychogonida (Arthropoda), with special reference to a microtubule-nuclear envelope complex. Acta Zoologica; in press
Fawcett, D. W.: The topographical relationship between the plane of the central pair of flagellar fibrils and the transverse axis of the head in Guinea-pig spermatozoa. J. Cell Sci. 3, 187–197 (1968)
Fawcett, D. W., Porter, K.: A study of the fine structure of ciliated epithelia. J. Morph. 94, 221–282 (1954)
Gibbons, I. R.: Chemical dissection of cilia. Arch. Biol. (Liège) 76, 317–352 (1965)
Gibbons, I. R., Fronk, E.: Some properties of bound and soluble dynein from sea urchin sperm flagella. J. Cell Biol. 54, 365–381 (1972)
Gibbons, B. H., Gibbons, I. R.: Flagellar movement and adenosine triphosphatase activity in sea urchin sperm extracted with triton x-100. J. Cell Biol. 54, 75–97 (1972)
Gibbons, I. R., Grimstone, A.: On flagellar structure in certain flagellates. J. biophys. biochem. Cytol. 7, 697–715 (1960)
Hopkins, J. M.: Subsidiary components of the flagella of Chlamidomonas reinhardii. J. Cell Sci. 7, 823–839 (1970)
Kiefer, B. I.: Development, organization, and degeneration of the Drosophila sperm flagellum. J. Cell Sci. 6, 177–194 (1970)
Markham, R., Frey, S., Hills, G. J.: Methods for the enhancement of image detail and accentuation of structure in electron microscopy. Virology 20, 88–102 (1963)
Ogawa, K., Mohri, H.: Studies on flagellar ATPase from sea urchin spermatozoa. Biochim. biophys. Acta (Amst.) 256, 142–155 (1972)
Phillips, D. M.: Insect sperm: their structure and morphogenesis. J. Cell Biol. 44, 243–277 (1970)
Ringo, D. L.: Flagellar motion and fine structure of the flagellar apparatus in Chlamidomonas. J. Cell Biol. 33, 543–571 (1967)
Stephens, R. E.: Thermal fractionation of outer fiber doublet microtubules into A- and B- subfiber components: A- and B- tubulin. J. molec. Biol. 47, 353–363 (1970)
Summers, K. E., Gibbons, I. R.: Effects of trypsin digestion on flagellar structures and their relationship to motility. J. Cell Biol. 58, 618–629 (1973)
Warner, F. D., Satir, P.: The substructure of ciliary microtubules. J. Cell Sci. 12, 313–326 (1973)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
van Deurs, B. Pycnogonid sperm. An example of inter- and intraspecific axonemal variation. Cell Tissue Res. 149, 105–111 (1974). https://doi.org/10.1007/BF00209053
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00209053