Abstract
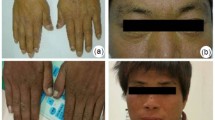
Erythropoietic protoporphyria (EPP), attributable to deficiency of ferrochelatase activity (FECH), is characterised mainly by cutaneous photosensitivity. To define the molecular defect in two EPP-affected siblings and their parents in a Swiss family, ferrochelatase cDNA was amplified by the polymerase chain reaction (PCR) and subjected to sequence analysis. A 5-bp deletion (T580G584) was identified on one allele of the ferrochelatase gene in both patients and their mother. Screening of the mutation among family members by RsaI digestion of PCR-amplified genomic DNA revealed autosomal dominant inheritance associated with abnormal protoporphyrin concentration and enzyme activity. We also isolated ferrochelatase cDNAs containing a 18-bp insertion (part of the intron 2 sequence) between exons 2 and 3; this corresponded to six extra amino acids (YESNIR) inserted between Arg-65 and Lys-66 of the known ferrochelatase. This isoform was identified initially in mRNAs derived from both alleles of the ferrochelatase gene in one patient. Its existence was confirmed in six additional EPP patients, in five out of seven controls, and in four different cell lines (fibroblast, muscle, hepatoma and myelogenous leukaemia). This isoform, roughly 20% of the total ferrochelatase mRNA, was generated through splicing at a second donor site in intron 2 and its presence was not linked to EPP.
Similar content being viewed by others
References
Brenner D, Didier J, Frasier F, Christensen S, Evans G, Dailey H (1992) A molecular defect in human protoporphyria. Am J Hum Genet 50:1203–1210
Lamoril J, Boulechfar S, Verneuil H de, Grandchamp B, Nordmann Y, Deybach J (1991) Human erythropoietic protoporphyria: two point mutations in the ferrochelatase gene. Biochem Biophys Res Commun 181:594–599
Nakahashi Y, Taketani S, Okuda M, Inoue K, Tolunaga R (1990) Molecular cloning and sequence analysis of cDNA encoding human ferrochelatase. Biochem Biophys Res Commun 173:748–755
Nakahashi Y, Fujita H, Taketani S, Ishida N, Kappas A, Sassa S (1992) The molecular defect of ferrochelatase in a patient with erythropoietic protoporphyria. Proc Natl Acad Sci USA 89:281–285
Nakahashi Y, Miyazaki H, Kadota Y, Naitoh Y, Inoue K, Yamamoto M, Hayashi N, Taketani S (1993a) Human erythropoietic protoporphyria: identification of a mutation at the splice donor site of intron 7 causing exon 7 skipping of the ferrochelatase gene. Hum Mol Genet 2:1069–1070
Nakahashi Y, Miyazaki H, Kadota Y, Naitoh Y, Inoue K, Yamamoto M, Hayashi N, Taketani S (1993b) Molecular defect in human erythropoietic protoporphyria with fatal liver failure. Hum Genet 91:303–306
Sarkany R, Alexander G, Cox T (1994) Recessive inheritance of erythropoietic protoporphyria with liver failure. Lancet 343: 1394–1396
Schneider-Yin X, Schäfer B, Möhr P, Burg G, Minder EI (1994) Molecular defect in erythropoietic protoporphyria with terminal liver failure. Hum Genet 93:711–713
Taketani S, Inazama J, Nakahashi Y, Abe T, Tokunaga R (1992) Structure of the human ferrochelatase gene exon/intron gene organization and location of the gene to chromosome 18. Eur J Biochem 205:217–222
Todd D, Hughes A, Ennis K, Ward A, Burrows D, Nevin N (1993) Identification of a single base pair deletion (40 del G) in exon 1 of the ferrochelatase gene in patient with erythropoietic protoporphyria. Hum Mol Genet 2:1495–1496
Wang X, Poh-Fitzpatrick M, Carriero D, Ostasiewicz L, Chen T. Taketani S, Piomelli S (1993) A novel mutation in erythropoietic protoporphyria: an aberrant ferrochelatase mRNA caused by exon skipping during RNA splicing. Biochim Biophys Acta 1181:198–200
Wang X, Poh-Fitzpatrick M, Taketani S, Chen T, Piomelli S (1994) Screening for ferrochelatase mutations: molecular heterogeneity of erythropoietic protoporphyria. Biochim Biophys Acta 1225:187–190
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schneider-Yin, X., Schäfer, B.W., Tönz, O. et al. Human ferrochelatase: a novel mutation in patients with erythropoietic protoporphyria and an isoform caused by alternative splicing. Hum Genet 95, 391–396 (1995). https://doi.org/10.1007/BF00208962
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00208962