Abstract
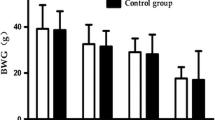
The one- and two-electron-reduction reactions of menadione result in the generation of reactive oxygen species which are believed to mediate the cytotoxicity of this xenobiotic. The induction of lipid peroxidation in liver and isolated hepatocytes occurs in response to the menadione-mediated formation of reactive oxygen species. However, studies on the effects of menadione on the urinary excretion of lipid metabolites have not been conducted. The effect of a single oral dose of 60 mg menadione/kg to rats on the urinary excretion of the lipid metabolites malondialdehyde (MDA), formaldehyde (FA), acetaldehyde (ACT), and acetone (ACON) has been examined over 48 h post-treatment. The urinary metabolites were identified by gas chromatography-mass spectrometry and quantitated by high pressure liquid chromatography. Time-dependent increases in the urinary excretion of the four metabolites were observed after menadione administration. Over the 48 h of the study, the menadione-induced urinary excretion of MDA, FA, ACT, and ACON increased by approximately 1.5-, 2.0-, 1.7-, and 3.2-fold, respectively, relative to control animals. The data were expressed in nmoles/kg body weight/4.5 h. The results clearly demonstrate that menadione increases the urinary excretion of four lipid metabolites. These metabolites may have widespread applicability as biomarkers of altered lipid metabolism in disease states and exposure to environmental pollutants/xenobiotics which induce enhanced lipid peroxidation. The non-invasive methods offer advantages over most other methods for assessing oxidative stress in vivo.
Similar content being viewed by others
References
Bagchi D, Bagchi M, Hassoun E, Stohs SJ (1992) Endrin-induced urinary excretion of formaldehyde, acetaldehyde, malondialdehyde and acetone in rats. Toxicology 75:81–389
—, —, —, — (1993a) Carbon tetrachloride-induced urinary excretion of formaldehyde, malondialdehyde, acetaldehyde and acetone in rats. Pharmacology 47:209–216
Bagchi D, Hassoun EA, Bagchi M, Stohs SJ (1993b) Protective effects of antioxidants against endrin-induced hepatic lipid peroxidation, DNA damage, and excretion of urinary lipid metabolites. Free Rad Biol Med 15:217–222
Bellomo G, Jewell SA, Thor H, Orrenius S (1982) Regulation of intracellular calcium compartmentation: Studies with isolated hepatocytes and t-butyl-hydroperoxide. Proc Natl Acad Sci USA 79:6842–6846
Bus JS, Gibson JE (1979) Lipid peroxidation and its role in toxicology. In: Hodgson et al. (eds) Reviews in biochemical toxicology. Elsevier, North-Holland, Amsterdam, pp 125–149
Comporti M (1989) Three models of free radical induced cell injury. Chem Biol Interact 72:1–56
Dhanakoti SN, Draper HH (1987) Response of urinary malondialdehyde to factors that stimulate lipid peroxidation in vivo. Lipids 22:643–646
Di Monte D, Ross D, Bellomo G, Eklow L, Orrenius S (1984a) Alterations in intracellular thiol homeostasis during the metabolism of menadione by isolated rat hepatocytes. Arch Biochem Biophys 235:334–342
Di Monte D, Bellomo G, Thor H, Nicotera P, Orrenius S (1984b) Menadione-induced cytotoxicity is associated with protein thiol oxidation and alteration in intracellular Ca2+ homeostasis. Arch Biochem Biophys 235:343–350
Foster DW (1987) Diabetes Mellitus. In: Braunwald et al. (eds) Harrison's Principles of internal medicine. McGraw-Hill, NY, pp 1778–1796
Iynagi T, Yamazaki I (1970) One-electron-transfer reactions in biochemical systems. V. Differences in the mechanism of quinone reduction by the NADH dehydrogenase and the NAD(P)H dehydrogenase (DT-diaphorase). Biochim Biophys Acta 216:282–294
Min K-S, Terano Y, Onosaka S, Tanaka K (1992) Induction of metallothionein synthesis by menadione or carbon tetrachloride is independent of free radical production. Toxicol Appl Pharmacol 113:74–79
Orrenius S, Ormstad K, Thor H, Jewell SA (1983) Turnover and function of glutathione studied with intact, isolated hepatic and renal cells. Fed Proc 42:3177–3188
Schulz H (1991) Beta oxidation of fatty acids. Biochim Biophys Acta 1081:109–120
Shara M, Dickson PH, Bagchi D, Stohs SJ (1992) Excretion of formaldehyde, malondialdehyde, acetaldehyde and acetone in the urine of rats in response to 2,3,7,8-tetrachlorodibenzo-p-dioxin, paraquat, endrin, and carbon tetrachloride. J Chromatogr 576:221–233
Slater TF (1984) Free radical mechanism of tissue injury. Biochem J 222:1–15
Thor H, Smith MT, Hartzell P, Bellomo G, Jewell SA, Orrenius S (1982) The metabolism of menadione (2-methyl-1,4-naphthoquinone) by isolated haptocytes. J Biol Chem 257:12,419–12,425
Warren DL, Reed DJ (1991) Modification of hepatic vitamin E stores in vivo. I. Alterations in plasma and liver vitamin E content by mathyl ethyl ketone peroxide. Arch Biochem Biophys 285:45–52
Winters DK, Klejan LA, Cederbaum AI (1988) Oxidation of glycerol to formaldehyde by rat liver microsomes. Biochem Biophys Res Commun 153:612–617
Yagi K (1987) Lipid peroxides and human diseases. Chem Phys Lipids 45:337–351
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bagchi, D., Moser, J. & Stohs, S.J. Quantitative determination of urinary lipid metabolites by high pressure liquid chromatography as indicators of menadione-induced in vivo lipid peroxidation. Arch. Environ. Contam. Toxicol. 26, 387–391 (1994). https://doi.org/10.1007/BF00203567
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00203567