Abstract
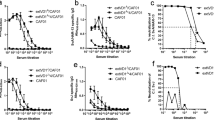
A human epithelioid cell line (A-431) was tested in parallel with McCoy fibroblast cells for the growth of trachoma-related serovar A Chlamydia trachomatis without centrifugation or cycloheximide addition. A-431 cells were 4–7 times more susceptible to infection with serovar A than McCoy cells in such unmanipulated cultures. Murine monoclonal antibodies (MAbs) developed against serovar A were then evaluated for their ability to inhibit unmanipulated serovar A infectivity of A-431 cells. Two of seven MAbs tested neutralized infectivity by more than 50%. An IgG2a MAb (2C8) that is specific for serovar A, and another IgG2a MAb (4E3) that reacts equally with serovars A and L2 neutralized infectivity of serovar A by 72.2 ± 3.7% and 56.0 ± 5.8% (mean ± SEM of 7 experiments) respectively. Mouse immune serum (MIS) raised against serovar A elementary bodies (EB) neutralized infectivity of serovar A by 76.0 ± 4.9% (mean ± SEM of 7 experiments). Immunoblot detection of serovar A EB polypeptides separated by SDS-PAGE indicated that 2C8 reacted with a 16 kD and 4E3 reacted with a 12 kD polypeptide while MIS reacted with several polypeptides including the major outer membrane protein (MOMP). These studies show that the human epithelioid cell line A-431 is a more susceptible host than McCoy cells in unmanipulated cultures, and that 2 MAbs neutralize serovar A infectivity of A-431 cells. Identification of antigenic moieties of importance in unmanipulated chlamydial infections may help in the development of potential vaccines against trachoma.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Caldwell HD, Perry LJ (1982) Neutralization of Chlamydia trachomatis infectivity with antibodies to the major outer membrane protein. Infect Immun 38:745–754
Colley DG, Goodman TA, Barsoum IS (1986) Ocular sensitization of mice by live (but not irradiated) Chlamydia trachomatis. Infect Immun 54:9–12
Fedorko DP, Clark RB, Nachamkin I, Dalton HP (1987) Complement dependent in vitro neutralization of Chlamydia trachomatis by subspecies-specific monoclonal antibody. Med Microbiol Immunol 176:225–228
Giard DJ, Aaronson SA, Todaro GJ, Arnstein P, Kersey JH, Dosik H, Parks WP (1973) In vitro cultivation of human tumors: Establishment of cell lines derived from a series of solid tumors. J Nat Cancer Inst 51:1417–1423
Goding SW (1976) Conjugation of antibody with fluorochromes: modification of the standard methods. J Immunol Meth 13:315–226
Hackstadt T (1986) Identification and properties of chlamydial polypeptides that bind eucaryotic cell surface components. J Bacteriol 165:13–20
Kearny JF, Radbruch A, Liesgang B, Rajewski K (1979) New mouse myeloma cell line that has lost immunoglobulin expression but permits the construction of antibody-secreting hybrid cell lines. J Immunol 123:1548
Kuo CC, Grayston JT (1976) Interaction of Chlamydia trachomatis organisms and HeLa 229 cells. Infect Immun 13:1103–1109
Kuo CC, Wang SP, Grayston JT (1973) Effect of polycations, polyanions and neuraminidase on the infectivity of trachoma-inclusion conjunctivitis and lymphogranuloma venereum organisms in HeLa cells: sialic acid residues as possible receptors for trachoma-inclusion conjunctivitis. Infect Immun 8:74–79
Lee CK (1981) Interaction between a trachoma strain of Chlamydia trachomatis and mouse fibroblasts (McCoy cells) in the absence of centrifugation. Infect Immun 31:584–591
Lucero ME, Kuo CC (1985) Neutralization of Chlamydia trachomatis cell culture infection by serovar-specific monoclonal antibodies. Infect Immun 50:595–597
Mergan DW, Stiver G, Peeling R, Maclean IW, Brunham RC (1988) Complement enhancement of neutralizing antibody to the structural protein of Chlamydia trachomatis. J Infect Dis 158:661–663
Moorman DR, Sixbey JW, Wyrick PB (1986) Interaction of Chlamydia trachomatis with human genital epithelium in culture. J Gen Microbiol 132:1055–1067
Peeling R, Maclean IA, Brunham RC (1984) In vitro neutralization of Chlamydia trachomatis with monoclonal antibody to an epitope on the major outer membrane protein. Infect Immun 46:484–488
Ripa KT, Mardh PA (1977) Cultivation of Chlamydia trachomatis in cycloheximide treated McCoy cells. J Clin Microbiol 6:328–331
Rota TR, Nichols RL (1973) Chlamydia trachomatis in cell culture. I. Comparison of efficencies of infection in several chemically defined media, at various pH and temperature values, and after exposure to diethylaminoethyldextran. Appl Microbiol 26:560–565
Thomas BJ, Evans RT, Hutchinson GR, Taylor-Robinson D (1977) Early detection of chlamydial inclusions combining the use of cycloheximide-treated McCoy cells and immunofluorescence staining. J Clin Microbiol 6:285–292
Towbin H, Stachelin T, Gordon J (1979) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: Procedure and some applications. PNAS 76:4350–4354
Wang SP (1971) A micro-immunofluorescence method. Study of antibody response to TRIC organisms in mice. In trachoma and related disorders caused by chlamydial agents. Nichols RL (ed) Excerpta Medica, Amsterdam, pp 273–288
Wards ME, Treharne JD, Murray A (1986) Antigenic specificity of human antibody to chlamydia in trachoma and lymphogranuloma venereum. J Gen Microbiol 132:1599–1610
Wenman WM, Meuser RU (1986) Chlamydia trachomatis elementary bodies possess proteins which bind to eucaryotic cell membranes. J Bacteriol 165:602–607
Zhang Y-X, Stewart S, Joseph T, Taylor HR, Caldwell HD (1987) Protecitve monoclonal antibodies recognize epitopes located on the major outer membrane protein of chlamydia trachomatis. J Immunol 138:575–581
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Barsoum, I.S., Goodman, T.A., Hardin, L.K. et al. Monoclonal antibody neutralization of unmanipulated Chlamydia trachomatis serovar A infection of human epithelioid cells (A-431). Med Microbiol Immunol 178, 113–120 (1989). https://doi.org/10.1007/BF00203307
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00203307