Abstract
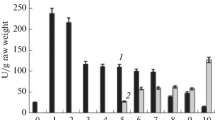
The subcellular localization and developmental changes of aconitase (EC 4.2.1.3) isoforms were studied in pumpkin tissues by anion-exchange chromatography. In etiolated pumpkin cotyledons three aconitase isoforms (denoted Aco I, II, and III, respectively) were found, and the first to be eluted (Aco I) represented the major part of the total aconitase activity in the tissue. Aconitase I gradually disappeared during the greening of cotyledons. In hypocotyls, roots and mature leaves Aco I was barely detectable, though the two other isoforms were present. In pumpkin mitochondria purified by differential or density gradient centrifugation, a high amount of Aco II was present, whereas the other isoforms were detected in low quantities. Since aconitase has never been clearly detected in glyoxysomal fractions, we conclude that the first and the third aconitase isoforms are cytosolic, while the second one represents the mitochondrial aconitase. Moreover, the first aconitase isoform is present in appreciable amounts only in etiolated cotyledons, so that it should represent the aconitase isoform involved in the glyoxylate cycle.
Similar content being viewed by others
Abbreviations
- Aco I (II, III):
-
aconitase isoform 1 (2, 3)
References
Breidenbach R.W., Beevers, H. (1967) Association of the glyoxylate cycle enzyme in a novel subcellular particle from castor bean endosperm. Biochem. Biophys. Res. Commun. 27, 462–469
Brouquisse, R., Nishimur M, Gaillard, J., Douce, R. (1987) Characterization of a cytosolic aconitase in higher plant cells. Plant Physiol. 84, 1402–1407
Constable, A., Quick, S., Gray, N.K., Hentze, M.W. (1992) Modulation of the RNA-binding activity of a regulatory protein by iron in vitro: switching between enzymatic and genetic function? Proc. Natl. Acad. Sci. USA 89, 4554–4558
Cooper, T.G., Beevers, H. (1969) Mitochondria and glyoxysomes from castor bean endosperm. J. Biol. Chem. 244, 3507–3513
Courtois-Verniquet, F., Douce, R. (1993) Lack of aconitase in glyoxysomes and peroxisomes. Biochem. J. 294, 103–107
De Bellis, L., Tsugeki, R., Alpi, A., Nishimura, M. (1993) Purification and characterization of aconitase isoforms from etiolated pumpkin cotyledons. Physiol. Plant. 88, 485–492
De Bellis L., Hayashi M., Biagi P.P., Hara-Nishimura, I., Alpi, A., Nishimura, M. (1994) Immunological analysis of aconitase in pumpkin cotyledons: the absence of aconitase in glyoxysomes. Physiol. Plant. 90, 757–762
Fansler, B., Lowenstain, J.M. (1969) Aconitase from pig heart. Methods Enzymol. 13, 26–30
Gerhardt, B.(1991) Peroxisomal catabolism of fatty and amino acid and its integration into cell metabolism. In: Molecular approaches to compartmentation and metabolic regulation, pp.121–128, Huang, A.H.C., Taiz, L., eds. The American Society of Plant Physiologists
Harris, H., Hopkinson, D.A. (1976) Handbook of electrophoresis in human genetics, pp. 4.2.1.3. Elsevier North Holland, Amsterdam
Huang, A.H.C., Trelease, R.N., Moore, T.S. (1983) Plant peroxisomes. Academic Press, New York
Kennedy, M.C., Mende-Muller, L., Blondin, G.A., Beinert, H. (1992) Purification and characterization of cytosolic aconitase from beef liver and its relationship to the iron-responsive element binding protein. Proc. Natl. Acad. Sci. USA 89, 11730–11734
Klausner, R.D., Rouault, T.A., Harford, J.B. (1993) Regulating of fate of mRNA: the control of cellular iron metabolism. Cell 72, 19–28
Laemmli, U.K. (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680–685
Mettler, I.J., Beevers, H. (1980) Oxidation of NADH in glyoxysomes by a malate-aspartate shuttle. Plant Physiol. 66, 555–560
Neuburger, M. (1985) Preparation of plant mitochondria, criteria for assessment of mitochondrial integrity and purity, survival in vitro. In: Encyclopedia of plant physiology — Higher plant cell respiration, vol. 18, pp. 7–24, Douce, R., Day, D.A., (eds.) Springer Verlag, Berlin
Rydén, L., Ofverstedt, L.G., Beinert, H., Emptage, M.H., Kennedy, M.C. (1984) Molecular weight of beef heart aconitase and stoichiometry of the components of its iron-sulfur cluster. J. Biol.Chem. 259, 3141–3144
Verniquet, F., Gaillard, J., Neuburger, M., Douce, R. (1991) Rapid inactivation of plant aconitase by hydrogen peroxide. Biochem. J. 84, 643–648
Yamaguchi, J., Nishimura, M. (1984) Purification of glyoxysomal catalase and immunochemical comparison of glyoxysomal and leaf peroxisomal catalase in germinating pumpkin cotyledons. Plant Physiol. 74, 261–267
Author information
Authors and Affiliations
Additional information
The authors thank Dr. Steven M. Smith (Institute of Cell and Molecular Biology, The University of Edinburgh, Edinburgh, UK) for critical reading of the manuscript. This work was supported in part by grants from a Grant-in-Aid for Scientific Research from the Ministry of Education, Science and Culture, Japan, and in part by the National Research Council of Italy (CNR, special project RAISA, subproject no. 2, paper no. 1580).
Rights and permissions
About this article
Cite this article
De Bellis, L., Hayashi, M., Nishimura, M. et al. Subcellular and developmental changes in distribution of aconitase isoforms in pumpkin cotyledons. Planta 195, 464–468 (1995). https://doi.org/10.1007/BF00202606
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00202606