Abstract
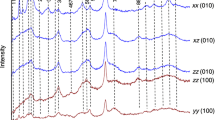
Polarized infrared absorption spectra of thin single-crystal slabs parallel to (010) and (001) of a staurolite from Pizzo Forno, Ticino, with analyzed composition (Fe2.9Mg0.9Zn0.1Mn0.1)Al17.5Ti0.1(Si7.7Al0.3)O48H3 have been measured in the range of 3000–4000 cm−1. From the pleochroitic behaviour of the OH-vibrations three groups of bands can be distinguished: the bands of group I, a strong band at 3445 cm−1 plus a weak shoulder at 3358 cm−1, and the bands of group II, a weak band centered at 3677 cm−1 plus a shoulder at 3635 cm−1, are assigned to the H1 and H2 protons, respectively. The bands of group III, a weak band at 3577 cm−1 plus a shoulder, cannot be interpreted on the basis of the proton positions known so far. We assign them to an additional proton H3, which is bonded to O1 and shows a bifurcated hydrogen bridge to two O5 in a vacant T2 site.
Similar content being viewed by others
References
Bellamy LJ, Owen AJ (1969) A simple relationship between the infra-red stretching frequencies and the hydrogen bond distance in crystals. Spectrochim Acta 25A:329–333
Fockenberg T (1992) Neue experimentelle Untersuchungen zur Stabilität von Pyrop und zum Chemismus von Mg-Staurolith im System MgO-Al2O3-SiO2-H2O(MASH). Ber Deutsche Min Ges 1:82
Hawthorne FC, Ungaretti L, Oberti R, Caucia F, Callegari A (1993) The crystal chemistry of staurolite. I. Crystal structure and site populations. Can Min 31:551–582
Holdaway MJ, Dutrow BL, Borthwick J, Shore P, Harmon RS, Hinton RW (1986) H content of staurolite as determined by H extraction line and ion microprobe. Am. Mineral. 71:1135–1141
Holdaway MJ, Mukhopadhyay B, Dyar MD, Rumble III D, Grambling JA (1991) A new perspective on staurolite crystal chemisty: Use of stoichiometric and chemical end-members for a mole fraction model. Am Mineral 76:1910–1919
Hornig DF, White HF, Reding FP (1958) The infrared spectra of crystalline H2O, D2O and HDO. Spectrochim Acta 12:338–349
Labotka TC, Rossman GR (1974) The infrared pleochroism of Lawsonite: The orientation of the water and hydroxide groups. Am Mineral 59:799–806
Langer K, Raith M (1974) Infrared spectra of Al-Fe(III)-Epidotes and Zoisites, Ca2(Al1- pFep/3+)Al2O(OH)[Si2O7][SiO4]. Am Mineral 59:1249–1258
Lonker SW (1983) The hydroxyl content of staurolite. Contrib. Mineral. Petrol 84:36–42
Nakamoto K, Margoshes M, Rundle RE (1955) Stretching frequencies as a function of distances in hydrogen bands. J Amer Chem Soc 77:6480–6488
Shinoda K, Aikawa N (1993) Polarized infrared absorbance spectra of an optically anisotropic crystal: Application to the orientation of the OH− dipole in quartz. Phys Chem Minerals 20: 308–314
Smith JV (1968) The crystal structure of staurolite. Am Mineral 53:1139–1155
Ståhl K, Kvick Å, Smith JV (1988) A neutron diffraction study of hydrogen positions at 13 K, domain model, and chemical composition of staurolite. J Sol State Chem 73:362–380
Tagai T, Joswig W (1985) Untersuchungen der Kationenverteilung im Staurolith durch Neutronenbeugung bei 100 K. N Jb Miner Mh 3:97–107
Takéuchi Y, Aikawa N, Yamamoto T (1972) The hydrogen locations and chemical composition of staurolite. Z Kristallogr 136:1–22
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koch-Müller, M., Langer, K. & Beran, A. Polarized single-crystal FTIR-spectra of natural staurolite. Phys Chem Minerals 22, 108–114 (1995). https://doi.org/10.1007/BF00202470
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00202470