Summary
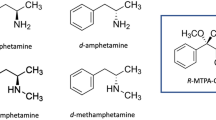
Optical isomers (d and l) and racemic compounds (dl) of methamphetamine (MAMP) and amphetamine (AMP), and biologic materials including those substances, could be analyzed by high performance liquid chromatography. Examining the temperature for the analysis, 40°C was the optimal condition in the reproducibility of separated MAMP-isomers. The reproducibility at the temperature did not vary significantly. The measured values of optical isomers were 0.116±0.012, 1.082±0.070 and 8.984±0.136 for the mixing ratios (l/d) of 0.111, 1.000, and 9.000, respectively. The detection limit for both d- and l-isomers was 25 ng.
The analytic result of hair specimens from two stimulant abusers by the present method indicates that they contained only d-MAMP and d-AMP, which is believed to have the strongest pharmacologic effect among the optical isomers of MAMP. The coefficient of variation in the analysis of five replicate standards, prepared by adding 1,000 ng each of racemate MAMP and AMP to hair, was less than 4%. The measured value against l/d=1.000 was 1.040±0.040 in MAMP and 0.980±0.030 in AMP. The detection limit for both racemate MAMP and AMP accumulated in hair was 250 ng.
The analysis of the optical isomers by our method would contribute to identifying the smuggling routes or the illicit method.
Zusammenfassung
Es wurde eine neue Methode zur Trennung von optisch aktiven Isomeren (d und l) und der racemischen Verbindung (dl) von Metamphetamin (MAMP) und Amphetamin (AMP) mittels Hochleistungsflüssigkeitschromatographie durch die UV-Methode ausgearbeitet. Wir fanden, daß 40°C in der Auflösung und der Reproduzierbarkeit der MAMP-Isomeren am besten waren. Meßwerte von Isomeren waren 0.116±0.012, 1.082±0.070 und 8.984±0.136 gegenüber den Mischungsverhältnissen (l/d) von 0.111, 1.000 und 9.000. Die Nachweisgrenze von d- und l-Isomeren war jeweils 25 ng.
Die Analysenergebnisse menschlicher Haare von zwei Stimulationsmittelkonsumenten zeigten mittels der oben genannten Methode, daß die Haare nur d-MAMP und d-AMP enthielten, welche unter den Isomeren die stärksten pharmakologischen Wirkungen haben. Die Streuung der Analysen von fünf Standard-Materialien, nach Zusatz von 1,000 ng jeder racemischen MAMP und AMP zu menschlichen normalen Kopfhaaren, war weniger als 4%; die Meßwerte von Isomeren gegenüber den Mischungsverhältnissen l/d=1.000, waren 1.040±0.040 bei MAMP und 0.980±0.030 bei AMP. Die Nachweisgrenze der racemischen Verbindung von MAMP und AMP, die in Haaren angehäuft sind, war jeweils 250 ng.
Die Analyse der Isomeren von MAMP und AMP durch unsere Methode würde der Aufdeckung von Schmuggelungen und -methoden dienen.
Similar content being viewed by others
References
Researches and Trainings Institutes of Ministry of Justice (1986) Crime by drug. Report of Crime in Japan in 1986, pp 27–33
Nagai N (1893) Studies on the components of Ephedraceae in herb medicine. Yakugaku Zasshi 139:901–933
Emde H (1929) Über Diastereomerie I. Konfiguration des Ephedrins. Helv Chim Acta 12:365–376
Rosenmund KW, Karg E (1942) Über die Darstellung von ß-Aryl-alkylaminen. Ber Dtsch Chem Ges 75B:1850–1859
Caldwell J, Dring LG, Williams RT (1972) Metabolism of 14C-methamphetamine in man, the guinea pig and the rat. Biochem J 129:11–22
Blackburn D, Burghard G (1965) Radiosynthesis of dextroamphetamine-14C sulfate. J Pharm Sci 54:1586–1588
Nagai T, Nagai T, Ikeda T (1984) Study on the drug abuse in forensic science. Detection of methamphetamine and amphetamine from hair, bone and teeth. In: Dunnet N, Kimber KJ (eds) TIAFT Proceedings of the 21st International Meeting 1984. MacMillan, New York Toronto London, pp 89–95
Frank RS (1983) The clandestine drug laboratory situation in the United States. J Forensic Sci 28:18–31
Novelli A (1939) Secondary amines by the Reuckart synthesis. J Am Chem Soc 61:520–521
Ogata A (1919) On the desoxyephedrine. Theory on structure of ephedrine. Yakugaku Zasshi 451:751–765
Kishi T, Inoue T, Suzuki S, Yasuda T, Oikawa T (1983) Analysis of impurities in methamphetamine. Eisei kagaku 29:400–406
Baron RP, Kruegel AV, Moore JM, Kram TC (1974) Identification of impurities in illicit methamphetamine samples. J Assoc Anal Chem 57:1147–1158
Gune M (1967) The urinary output d- and l-amphetamine. Biochem Pharamacol 16:863–869
Gordis E (1966) Gas-chromatographic resolution of optical isomers in microgram samples of amphetamine. Biochem Pharmacol 15:2124–2126
Niwaguchi T, Kanda Y, Kishi T, Inoue T (1982) Determination of d-methamphetamine in urine after administration of d- or dl-methamphetamine to rats by radioimmunoassay using optically sensitive antiserum. J Forensic Sci 27:592–597
Baumgartner AM, Jones PF, Baumgartner WA, Black CT (1979) Radioimmunoassay of hair determining opiate-abuse histories. J Nuclear Med 20:748–752
Klug E (1980) Zur Morphinbestimmung in Kopfhaaren. Z Rechtsmed 84:183–193
Dring LG, Smith RL, Williams RT (1970) The metabolic fate of amphetamine in man and other species. Biochem J 116:425–435
Beckett AH, Rowland M (1965) Urinary excretion kinetics of methylamphetamine in man. J Pharm Pharmacol 17:109s-114s
Ishiyama I, Nagai T, Toshida S (1983) Detection of basic drugs (methamphetamine, antidepressants, and nicotine) from human hair. J Forensic Sci 28:380–385
Niwaguchi T, Suzuki S, Inoue T (1983) Determination of methamphetamine in hair after single and repeated administration to rat. Arch Toxicol 52:157–164
Suzuki S, Inoue T, Yasuda T (1984) Analysis of methamphetamine in hair, nail, saliva and sweat by mass fragmentography. In: Dunnett N, Kimber KJ (eds) TIAFT Proceedings of the 21st International Meeting 1984. MacMillan, New York Toronto London, pp 95–100
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nagai, T., Kamiyama, S. Forensic toxicologic analysis of methamphetamine and amphetamine optical isomers by high performance liquid chromatography. Z Rechtsmed 101, 151–159 (1988). https://doi.org/10.1007/BF00201110
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00201110