Summary
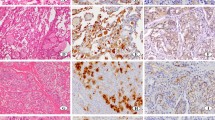
The activities of A, B and H in serous cells (S-cells), mucous cells (M-cells) and excretory duct cells were examined in a large number of paraffin sections of three major salivary glands obtained from 91 corpses, using the immunofluorescence technique. The results are:
-
1.
By taking H activity in S-cells of the submandibular gland or A, B and H activity in M-cells of the sublingual gland as an indicator, the salivary glands were classified as Type I showing activity and Type II showing no activity. No glands corresponding to the intermediate type, as seen in the case of saliva, were noted at all. Among 91 corpses, 70 cases were classified as Type I and 21 as Type II. The results matched well with those of Lewis type tested on blood. The frequencies of the typing (Type I; 76.9%, Type II; 23.1%) were approximately in concordance with those of secretor and nonsecretor in Japanese saliva. From these results, it was assessed that the former corresponded to the secretor type in the case of saliva, and the latter to the nonsecretor type.
-
2.
Even in the same individual, both S-cells and M-cells exhibited different productivities of substances, depending on the glands to which they belonged. Namely, only S-cells in the submandibular gland belonging to Type I showed only H activity independent of the blood group of the individual, but the other S-cells in the other major glands did not show any activity for A, B and H. M-cells exhibited strong activity for H and/or A and/or B in the sublingual and submandibular gland and belonged to Type I, but little activity in the sublingual gland belonged to Type II. In the submandibular gland of Type II, some M-cells showed activity and others did not.
-
3.
On the basis of the above results, we discuss the applicability of the present genetic theory concerning the secretor and nonsecretor type in saliva to salivary glands and cells, and further refer to the reasons for appearance of the weak secretor type or intermediate type in saliva.
Zusammenfassung
Es wurde die A-, B- und H-Aktivität der serösen (S), der mukösen (M) und der Ausführungsgangszelle von aus 91 Leichen entnommen und in Paraffin eingebetteten Hauptspeicheldrüsen mittels des Immunofluoreszenzverfahrens untersucht.
Die Speicheldrüsen wurden in zwei Typen eingeteilt: in den aktiven Typ I und den inaktiven Typ II; die Submandibularis mit der H-Aktivität der S-Zellen und die Sublingualis mit der ABH-Aktivität der M-Zellen. Ein Zwischentyp wie beim Speichel wurde nicht beobachtet. Die Gruppierung und die Typisierung der Speicheldrüsen stimmten mit dem ABO- und Lewis-System der zugehörigen Blute überein. Unter den 91 Leichen gehörten 70 zum Typ I (76,9%) und 21 zum Typ II (23,1%). Diese Frequenzen waren in Einklang mit dem einschlägigen Befund (Ausscheider bzw. Nichtausscheider) bei den Japanern.
Unabhängig vom Individuum wiesen die S- und M-Zellen der zugehörigen Speicheldrüsen variable Aktivitäten auf. Die S-Zellen des Typs I der Submandibularis erzeugten nur H-Aktivität, die von der Blutgruppe unabhängig war, während die übrigen S-Zellen der Hauptspeicheldrüsen keine Aktivität zeigten. Die zum Typ I gehörenden M-Mandibularis- und Sublingualiszellen zeigten eine starke H- und A- und/oder B-Aktivität. Beim Typ II erzeugten nur ein Teil der M-Zellen der Submandibularis- und Sublingualisdrüse H- und A- und/oder B-Aktivität.
Aufgrund dieser Ergebnisse wurde die Wechselbeziehung zwischen der Typisierung der Speicheldrüsen und der des Speichels, die Gültigkeit der gegenwärtigen genetischen Theorie über Sekretor und Nichtsekretor im Speichel sowie die Ursache für das Auftreten des schwachen Sekretors oder des Zwischentyps erörtert.
Similar content being viewed by others
References
Yamakami K (1926) The individuality of semen with reference to its property of inhibiting specifically isohemoagglutination. J Immunol 12:185–189
Landsteiner K, Levine P (1926) On group specific substances in human spermatozoa. J Immunol 12:415–418
Schiff F, Sasaki H (1932) Der Ausscheidungstypus, ein auf serologischem Wege nachweisbares Mendelndes Merkmal. Klin Wochenschr 11:1426–1429
Race RR, Sanger R (1975) Blood groups in man (6th ed) Blackwell scientific publications, London, pp 311–322
Grubb R (1948) Correlation between Lewis blood group and secretor charactor in man. Nature 162:933
Grubb R, Morgan WTJ (1949) The Lewis blood group characters of erythrocytes and body-fluids. Brit J Exp Pathol 30:198–208
Grubb R (1951) Observation on the human group system Lewis. Acta Pathol Microbiol Scand 28:61–81
Fotino M, Aloman S (1959) Diagnose des Ausscheidertypus mit pflanzlichem Anti-H (Evonymus europaea). Folia haematol (Frankfurt) 3:227–235
Clarke CA, McConnell RB, Sheppard PM (1960) A genetical study of the variations in ABH secretion. Ann Hum Genet 24:295–307
McNeil C, Trentelman EF, Kreutzer VO, Fullmer CD (1957) Aberrant secretion of salivary A, B and H group substances in human beings. Amer J Clin Pathol 28:145–151
Bhatia HM, Randeria KJ (1970) Studies on blood group antigens in saliva: Incidence and type of aberrant secretors. Indian J Med Res 58:194–201
Williams CA, Chase MW (1976) Method in immunology and immunochemistry, V. Academic press, New York, pp 425–433
Coons AH, Kaplan MH (1950) Localization of antigen in tissue cells. J Exp Med 91:1–13
Rygaard J, Olsen W (1969) Interference filters for improved immunofluorescence microscopy. Acta Pathol Microbiol Scand 76:146–148
Furuhata T (1974) Development of haemotypology in Japan. Tokyo standard serum Ltd, Tokyo, p 98
Glynn LE, Holborow EJ (1959) Distribution of blood-group substances in human tissues. Brit Med Bull 15:150–153
Holborow EJ, Brown PC, Glynn LE, Hawes MD, Gresham GA, O'brien TF, Coombs RRA (1960) The distribution of the blood group A antigen in human tissues. Brit J Exp Pathol 41:430–437
Szulman AE (1960) The histological distribution of blood group substances A and B in man. J Exp Med 111:785–807
Szulman AE (1962) The histological distribution of the blood group substances in man as disclosed by immunofluorescence. J Exp Med 115:977–996
Davidsohn I (1972) Early immunologic diagnosis and prognosis of carcinoma. Am J Clin Pathol 57:715–730
Shen L, Grollman EF, Ginsburg V (1968) An enzymatic basis for secretor status and blood group substance specificity in humans. Proc NAS 59:224–230
Chester MA, Watkins WM (1969) α-L-fucosyltransferases in human submaxillary gland and stomach tissues associated with the H, Lea and Leb blood-group characters and ABH secretor status. Biochem Biophys Res Commun 34:835–842
Watkins WM (1966) Blood-group substances, in the ABO system the genes control the arrangement of sugar residues that determines blood-group specificity. Science 152:172–181
Morgan WTJ, Watkins WM (1969) Genetic and biochemical aspects of human blood-group A-, B-, H-, Lea- and Leb-specificity. Br Med Bull 30–34
Schneyer LH (1956) Source of resting total mixed saliva of man. J Appl Physiol 9:79–81
Kerr AC (1961) The physiological regulation of salivary secretion in man. International Series of Monographs on Oral Biology. Pergamon Press, Oxford, p 9
Dawes C, Wood CM (1973) The contribution of oral minor mucous gland secretions to the volume of whole saliva in man. Archs Oral Biol 18:337–342
Milne RW, Dawes C (1973) The relative contributions of different salivary glands to the blood group activity of whole saliva in humans. Vox Sang 25:298–307
Hensten-Pettersen A, Kornstad L (1976) The contribution of the minor mucous glands to the concentrations of blood group specific substances, carbohydrates and proteins in human mixed saliva. Archs Oral Biol 21:485–489
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Takahashi, M., Kamiyama, S. Immunohistological studies on ABH-activities in secretory cells of human major salivary glands—correlation between ABH-activities in the secretory cells and secretor-nonsecretor. Z Rechtsmed 95, 217–226 (1985). https://doi.org/10.1007/BF00200455
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00200455