Abstract
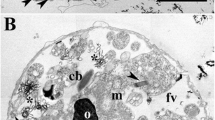
The results of studies of Micrasterias rotata (Grev.) Ralfs, M. thomasiana Archer (biradiate and uniradiate forms) and Closterium sp. using one- and two-dimensional vibrating probes show that transcellular ionic currents are detectable only around cells undergoing expansion of the primary cell wall (half-cell); current enters local regions of expansion and exits over both the rigid surface of the secondary wall and regions of the primary wall where hardening of the wall prevents further expansion. Current densities remain at steady levels until expansion stops with maturation of the primary wall, whereupon currents are no longer detectable. The temporal and spatial correlation between the currents and regions of wall expansion is particularly evident because morphogenesis of the half-cell is a determinate process. Measurements of inward currents ranged from 0.1 to 5.4 μA · cm−2, and outward currents ranged from-0.05 to -1.5 μA · cm−2 measured at 18 μ from the cell surface. The results of ion substitution and channel-blocker studies indicate that the currents may be carried at least in part by Ca2+, Cl−, H+ and K+ ions. The possible role of a Ca2+ influx during tip growth in desmids is discussed.
Similar content being viewed by others
References
Brawley, S.H., Robinson, K.R. (1985) Cytochalasin treatment disrupts the endogenous currents associated with cell polarization in fucoid zygotes: studies of the role of F-actin in embryogenesis. J. Cell Biol. 100, 1173–1184
Brower, D.L., Giddings, T. (1980) The effects of applied electric fields on Micrasterias. II. The distributions of cytoplasmic and plasma membrane components. J. Cell Sci. 42, 279–290
Brower, D.L., McIntosh, J.R. (1980) The effects of applied electric fields in Micrasterias. II. The distributions of cytoplasmic and plasma membrane components. J. Cell Sci. 42, 279–290
Doucherty, R.J. (1988) Gadolinium selectively blocks a component of the calcium current in rodent neuroblastoma. J. Physiol. (Lond.) 398, 33–47
Hepler, P.K., Wayne, R.O. (1985) Calcium and plant development. Annu. Rev. Plant Physiol. 36, 397–439
Jaffe, L.F., Nuccitelli, R. (1974) An ultrasensitive vibrating probe for measuring steady extracellular currents. J. Cell Biol. 63, 614–628
Jaffe, L.F., Robinson, K.R., Nuccitelli, R. (1974) Local cation entry and self-electrophoresis as an intracellular localization mechanism. Ann. N.Y. Acad. Sci. 238, 372–389
Kallio, P., Lehtonen, J. (1981) Nuclear control of cytomorphogenesis in Micrasterias. In: Cytomorphogenesis in plants (Cell Biology Monogr. No. 8), pp. 191–213, Kiermayer, O., ed. Springer, New York Berlin Heidelberg
Kiermayer, O. (1981) Cytoplasmic basis of morphogenesis in Micrasterias. In: Cytomorphogenesis in plants (Cell Biol. Monogr. No. 8), pp. 147–188, Kiermayer, O., ed. Springer, New York Berlin Heidelberg
Kiermayer, O., Meindl, U. (1989) Cellular morphogenesis: the desmid (Chlorophyceae) system. In: Algae as experimental systems, pp. 149–167, Coleman, A.W., Goff, L.J., Stein-Taylor, J.R., eds. Liss, New York
Lacalli, T.C. (1975a) Morphogenesis in Micrasterias. I. Tip growth. J. Embryol. Exp. Morphol. 33, 95–115
Lacalli, T.C. (1975b) Morphogenesis in Micrasterias. II. Patterns of morphogenesis. J. Embryol. Exp. Morphol. 33, 117–126
Lacalli, T.C., Acton, A.B. (1974) Tip growth in Micrasterias. Science 183, 665–666
Lehtonen, J. (1984) The significance of Ca2+ in the morphogenesis of Micrasterias studied with EGTA, verapamil, LaCl3 and calcium ionophore A23187. Plant Sci. Lett. 33, 53–60
McKerracher, L.J., Heath, I.B. (1987) Cytoplasmic migration and intracellular organelle movements during tip growth of fungal hyphae. Exp. Mycology 11, 79–100
McNally, J.G., Cowan, J.D., Swift, H. (1983) The effects of the ionophore A23187 on pattern formation in the alga Micrasterias. Devel. Biol. 97, 137–145
Meindl, U. (1982) Local accumulation of membrane-associated calcium according to cell pattern formation in Micrasterias denticulata, visualized by chlorotetracycline fluorescence. Protoplasma 110, 143–146
Nawata, T. (1988) Effects of ions and membrane potential on the elongation of the unicellular green alga Closterium. Plant Cell Physiol. 29, 951–959
Oberliethner, H., Ritter, M., Lang, F., Guggins, W. (1983) Anthracene-9-carboxylic acid inhibits renal chloride reabsorption. Pflügers Arch. 398, 172–174
Palade, P.T., Barchi, R.L. (1977) On the inhibition of muscle membrane chloride conductance by aromatic carboxylic acids. J. Gen. Physiol. 69, 879–896
Pickett-Heaps, J.D. (1983) Morphogenesis in desmids: our present state of ignorance. Modern Cell Biol. 2, 241–258
Scheffey, C. (1986) Pitfalls of the vibrating probe technique, and what to do about them. In: Ionic currents in development, pp. 3–12, Nuccitelli, R., ed. Liss, New York
Scheffey, C. (1988) Two approaches to construction of vibrating probes for electrical current measurement in solution. Rev. Sci. Instr. 59, 787–792
Starr, R.C. (1964) The culture collecton of algae at Indiana University. Am. J. Bot. 51, 1013–1044
Steer, M.W. (1988) The role of calcium in exocytosis and endocytosis in plant cells. Physiol. Plant. 72, 213–220
Troxell, C.L. (1988) Transcellular ionic currents in the algae Acetabularia, Micrasterias and Closterium. Ph.D. Thesis, University of Colorado, Boulder, USA. Dissertation Abst. Internat. 49, 2977-B, Abstr. No. DA8819710
Troxell, C.L., Scheffey, C., Pickett-Heaps, J.D. (1986) Ionic currents during wall morphogenesis in Micrasterias and Closterium. In: Ionic currents in development, pp. 105–112, Nuccitelli, R., ed. Liss, New York
Ueda, K., Noguchi, T. (1988) Microfilament bundles of F-actin and Cytomorphogenesis in the green alga Micrasterias crux-melitensis. Eur. J. Cell Biol. 46, 61–67
Waris, H. (1953) The significance for algae of chelating substances in nutrient solutions. Physiol. Plant. 6, 538–543
Wehr, J.D., Brown, L.M., Vandereest, I.E. (1986) Hydrogen ion buffering capacity of culture media for algae from moderately acidic, oligotrophic waters. J. Phycol. 22, 88–94
Weisenseel, M.H., Kircherer, R.M. (1981) Ionic currents as control mechanisms in Cytomorphogenesis. In: Cytomorphogenesis in plants (Cell Biol. Monogr. No. 8), pp. 379–399, Kiermayer, O., ed. Springer, New York Berlin Heidelberg
Author information
Authors and Affiliations
Additional information
This work was conducted at the National Vibrating Probe Facility, Marine Biological Laboratory, Woods Hole, Mass., USA. Dr. Lionel F. Jaffe, Director of the Facility, and Dr. Jeremy D. PickettHeaps, University of Colorado, Boulder, USA, provided valuable guidance and support, and gave unstinting encouragement during these studies. Dr. Franklin M. Harold provided support for the writing of this paper during C.L.T.'s postdoctoral year at the National Jewish Center for Immunology and Respiratory Research, Denver. Mr. Alan Shipley and Mr. Steve Dixon provided talented technical assistance. C.L.T. is grateful for support received from a National Institutes of Health Pre-doctoral Training Grant in the Department of Molecular, Cellular and Developmental Biology, University of Colorado. The work was supported by N.I.H. grants 5 P41 RR01395 and 3 P41 RR01395-02S1 (to L.F.J.), National Science Foundation grants No. BSR 82 14199 and PCM 83 09331 (to J.P.-H.), and No. DCB 86 18694 (to F.M.H.).
Rights and permissions
About this article
Cite this article
Troxell, C.L., Scheffey, C. Ionic currents flow through Micrasterias and Closterium cells during expansion of the primary cell wall. Planta 184, 218–225 (1991). https://doi.org/10.1007/BF00197950
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00197950