Abstract
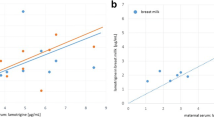
Nalbuphine, a mixed agonist-antagonist opiate, is commonly used as a systemic analgesic during labour. Recent reports of perinatal adverse effects prompted us to carry out therapeutic nalbuphine monitoring in obstetric analgesia. Because data on fetomaternal transfer are scarce and the pharmacokinetics of this drug in the neonate are largely unknown, we report data obtained from 28 parturients treated with nalbuphine either intramuscularly and/or intravenously during labour. Plasma nalbuphine levels were measured by high-performance liquid chromatography (HPLC) with electrochemical detection. At delivery, 30–150 min after maternal administration, nalbuphine concentrations ranged from 5.0 to 79.2 ng·ml−1 in mother plasma samples and from 3.0 to 46.6 ng·ml−1 in umbilical cord plasma samples. Nalbuphine concentrations were highly correlated to dose. The fetomaternal ratio was high: 0.74 and not correlated to the administered dose of nalbuphine. An estimated plasma half-life of 4.1 h was calculated from two determinations in the neonate based on the assumption of a monoexponential decay of nalbuphine concentrations. Apart from a flattening of the fetal heart rate tracing in 54% of the cases, only one neonate had a low Apgar score at birth. The apparent prolonged half-life of nalbuphine in the neonate indicates the usefulness of an intramuscular injection of naloxone to prevent recurrence of cardiorespiratory depression due to nalbuphine administration to the mother.
Similar content being viewed by others
References
Dadabhoy ZP, Tapia DP, Zsigmond EK (1985) Transplacental transfer of nalbuphine in patients undergoing cesarean section. Anesth Analg 64:205
Dan U, Rabinovici Y, Barkai G, Modan M, Etchin A, Mashiach S (1991) Intravenous pethidine and nalbuphine during labor: a prospective double-blind comparative study. Gynecol Obstet Invest 32:39–43
Errick JK, Heel RC (1983) Nalbuphine: a preliminary review of its pharmacological properties and therapeutic efficacy. Drugs 26:191–211
Feinstein SJ, Lodeiro JG, Vintzileos AM, Campbell WA, Montgomery JT, Nochimson DJ (1986) Sinusoidal fetal heart rate pattern after administration of nalbuphine hydrochloride: a case report. Am J Obstet Gynecol 154:159–160
Franck M, McAteer EJ, Cattermole R, Loughnan B, Stafford LB, Hitchcock AM (1987) Nalbuphine for obstetric analgesia: a comparison of nalbuphine with pethidine for pain relief in labour when administered by patient-controlled analgesia. Anaesthesia 42:697–703
Guillonneau M, Jacqz-Aigrain E, De Crepy A, Zeggout H (1990) Perinatal adverse effects of nalbuphine given during parturition. Lancet 335:1588
Hodgkinson R, Husain FJ (1982) The duration of effect of maternally administered meperidine on neonatal neurobehaviour. Anesthesiology 56:51–52
Jaillon P, Gardin ME, Lecocq B, Richard MO, Meignan S, Blondel Y, Grippat JC, Bergnieres J, Vergnoux O (1989) Pharmacokinetics of nalbuphine in infants, young healthy volunteers and elderly patients. Clin Pharmacol Ther 46:226–233
Martin CB (1982) Physiology and cinical use of fetal heart rate variability. In: Petrie RH (ed) Clinics in perinatology, symposium on fetal monitoring, vol 9, No. 2. WB Saunders, Philadelphia, pp 339–352
Miller RR (1980) Evaluation of nalbuphine hydrochloride. Am J Hosp Pharm 37:942–949
Miller JW, Anderson HH (1954) The effect of N-demethylation on certain pharmacological actions of morphine, codeine and meperidine in the mouse. J Pharmacol Exp Ther 112:191–196
Milne B, Jhamandas K (1984) Naloxone: new therapeutic roles. Can Anaesth Soc J 31:272–278
Moreland TA, Brice JEH, Walker CHM, Parija AC (1980) Naloxone pharmacokinetics in the newborn. Br J Clin Pharmacol 9:609–612
Morselli PL, Rovei V (1980) Placental transfer of pethidine and norpethidine and their pharmacokinetics in the newborn. Eur J Clin Pharmacol 18:25–30
Morselli PL, Franco-Morselli R, Bossi L (1980) Clinical pharmacokinetics in newborns and infants. Age related differences and therapeutic implications. Clin Pharmacokinet 5:485–527
Nicolle E, Michaud S, Serre-Debeauvais F, Bessard G (1995) A rapid and sensitive high-liquid chromatographic assay for nalbuphine in plasma. J Chromatogr B 663:111–117
Romagnoli A, Keats AS (1980) Ceiling effect for respiratory depression by nalbuphine. Clin Pharmacol Ther 27:478–485
Sear JW, Keegan M, Kay B (1987) Disposition of nalbuphine in patients undergoing general anaesthesia. Br J Anaesth 59: 572–575
Sgro C, Escousse A, Tennenbaum D, Gouyon JB (1990) Perinatal adverse effects of nalbuphine given during labour. Lancet 336:1070
Stile IL, Fort M, Warzburger RJ, Rodvold KA, Spector S, Hiatt IM, Hegyi T (1987) The pharmacokinetics of naloxone in the premature newborn. Dev Pharmacol Ther 10:454–459
Wilson CM, Dundee JW, McClean E, Moore J (1985) Transplacental gradient of pethidine and nalbuphine in labour. Proceedings of the BPS, 18–20 December 1985, pp 571–572
Wilson CM, McClean E, Moore J, Dundee JW (1986a) A double-blind comparison of intramuscular pethidine and nalbuphine in labour. Anaesthesia 41:1207–1213
Wilson SJ, Errick JK, Balkon J (1986b) Pharmacokinetics of nalbuphine during parturition. Am J Obstet Gynecol 155:340–344
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nicolle, E., Devillier, P., Bessard, G. et al. Therapeutic monitoring of nalbuphine: transplacental transfer and estimated pharmacokinetics in the neonate. Eur J Clin Pharmacol 49, 485–489 (1996). https://doi.org/10.1007/BF00195935
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00195935