Abstract
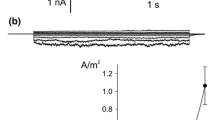
The characteristics of Cl− transport in isolated tonoplast vesicles from red-beet (Beta vulgaris L.) storage tissue have been investigated using the Cl−-sensitive fluorescent probe, 6-methoxy-1-(3-sulfonatopropyl)-quinolinium (SPQ). The imposition of (inside) positive diffusion potentials, generated with K+ and valinomycin, increased the initial rate of Cl− transport, demonstrating that Cl− could be electrically driven into the vesicles. Chloride influx was unaffected by SO 2-4 , but was competitively blocked by NO −3 , indicating that both Cl− and NO −3 may be transported by the same porter. In some preparations, increases in free-Ca2+ concentration from 10−8 to 10−5 mol·dm−3 caused a significant decrease in Cl− influx, which may indicate that cytosolic Ca2+ concentration has a role in controlling Cl− fluxes at the tonoplast. However, this effect was only seen in about 50% of membrane preparations and some doubt remains over its physiological significance. A range of compounds known to block anion transport in other systems was tested, and some partially blocked Cl− transport. However, many of these inhibitors interfered with SPQ fluorescence and so only irreversible effects could be tested. The results are discussed in the context of recent advances made using the patch-clamp technique on isolated vacuoles.
Similar content being viewed by others
Abbreviations
- BTP:
-
1,3-bis[tris(hydroxymethyl)-methylamino]propane
- DTT:
-
dithiothreitol
- EDTA:
-
ethylenediaminetetraacetic acid
- Δψ :
-
membrane potential
- ΔpH:
-
pH gradient
- SPQ:
-
6-methoxy-1-(3-sulfonatopropyl)quinolinium
- Tricine:
-
N-[2-hydroxy-1,1-bis(hydroxymethyl)ethyl] glycine
References
Bates, G.W., Goldsmith, M.H.M., Goldsmith, T.H. (1982) Separation of tonoplast and plasma membrane potential and resistance in cells of oat coleoptiles. J. Membr. Biol. 66, 15–23
Bennett, A.B., Spanswick, R.M. (1983) Optical measurement of ΔpH and Δψ in corn root membrane vesicles: kinetic analysis of Cl− effects on a proton-translocating ATPase. J. Membr. Biol. 71, 95–107
Blumwald, E., Poole, R.J. (1985) Na+/H+ antiport in isolated tonoplast vesicles from storage tissue of Beta vulgaris L. Plant Physiol. 78, 163–167
Bradford, M.M. (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilising the principle of dye-binding. Anal. Biochem. 72, 248–256
Bretag, A.H. (1987) Muscle chloride channels. Physiol. Rev. 67, 621–724
Bretag, A.H., Dawe, S.R., Kerr, D.I.B., Moskwa, A.G. (1980) Myotonia as a side effect of diuretic action. Br. J. Pharmacol. 71, 467–471
Brule, G., Hardecour, G., Idaiha, H., Guilbault, P. (1983) Inhibition par une substance amphiphile, l'acide niflumique, de la rectification dans le sens entrant de la fibre musculaire de crustacea. Arch. Int. Physiol. Biochem. 91, 269–277
Cabantchik, Z.I., Knauf, P.A., Rothstein, A. (1978) The anion transport system of the red blood cell. The role of membrane protein evaluated by the use of ‘probes’. Biochim. Biophys. Acta 515, 239–302
Chen, P.Y., Verkman, A.S. (1988) Sodium-dependent chloride transport in basolateral membrane vesicles isolated from rabbit proximal tubule. Biochemistry 27, 655–660
Chen, P.Y., Illsley, N.P., Verkman, A.S. (1988) Renal brush border chloride transport mechanisms characterised using a fluorescent indicator. Am. J. Physiol. 254, F114-F120
Churchill, K.A., Sze, H. (1984) Anion-sensitive, H+-pumping ATPase of oat roots. Direct effects of Cl−, NO -3 , and a disulfonic stilbene. Plant Physiol. 87, 490–497
Coyaud, L., Kurkdjian, A., Kado, R., Hedrich, R. (1987) Ion channels and ATP-driven pumps involved in ion transport across the tonoplast of sugarbeet vacuoles. Biochim. Biophys. Acta 902, 263–268
Cupoletti, J., Sachs, G. (1986) Regulation of gastric acid secretion via modulation of a chloride conductance. J. Biol. Chem. 23, 14952–14959
Dechecci, M.C., Cabrini, G. (1988) Chloride conductance in membrane vesicles from human placenta using a fluorescent probe. Implications for cystic fibrosis. Biochim. Biophys. Acta 945, 113–120
Falke, J.J., Chan, S.I. (1986a) Molecular mechanisms of band 3 inhibitors. 1. Transport site inhibitors. Biochemistry 25, 7888–7894
Falke, J.J., Chan, S.I.. (1986b) Molecular mechanisms of band 3 inhibitors. 2. Channel blockers. Biochemistry 25, 7895–7898
Falke, J.J., Chan, S.I. (1986c) Molecular mechanisms of band 3 inhibitors. 3. Translocation inhibitors. Biochemistry 25, 7899–7906
Flowers, T.J.. (1988) Chloride as a nutrient and as an osmoticum. In: Advances in plant nutrition, vol. 3, pp. 55–78, Tinker, P.B., Läuchli, A., eds. Praeger, New York
Garlid, K.D., Beavis, A.D. (1986) Evidence for the existence of an inner membrane anion channel in mitochondria. Biochim. Biophys. Acta 853, 187–204
Hedrich, R., Kurkdjian, A. (1988) Characterisation of an anionpermeable channel from sugar beet vacuoles: effect of inhibitors. EMBO J. 7, 3661–3666
Hedrich, R., Neher, E. (1987) Cytoplasmic calcium regulates voltagedependent ion channels in plant vacuoles. Nature 329, 833–836
Hedrich, R., Barbier-Brygoo, H., Felle, H., Flügge, U.I., Lüttge, U., Maathuis, F.J.M., Marx, S., Prins, H.B.A., Raschke, K., Schnabl, H., Schroeder, J.I., Struve, I., Taiz, L., Zeigler, P. (1988) General mechanisms for solute transport across the tonoplast of plant vacuoles: a patch-clamp survey of ion channels and proton pumps. Bot. Acta 101, 7–13
Illsley, N.P., Verkman, A.S. (1987) Membrane chloride transport measured using a chloride-sensitive fluorescent probe. Biochemistry 26, 1215–1219
Jennings, I.R., Rea, P.A., Leigh, R.A., Sanders, D. (1988) Quantitative and rapid estimation of H+ fluxes in membrane vesicles: Software for analysis of fluorescence quenching and relaxation. Plant Physiol. 86, 1257–1263
Kaestner, K.H., Sze, H. (1987) Potential-dependent anion transport in tonoplast vesicles from oat roots. Plant Physiol. 83, 483–489
Lunevsky, V.Z., Zherelova, O.M., Vostrikov, I.Y., Berestovsky, G.N. (1983) Excitation of characean cell membranes as a result of activation of calcium and chloride channels. J. Membr. Biol. 72, 43–58
MacRobbie, E.A.C. (1970) The active transport of ions in plant cells. Quart. Rev. Biophys. 3, 251–294
Nelson, D.J., Tang, J.M., Palmer, L.G. (1984) Single-channel recordings of apical membrane chloride conductance in A6 epithelial cells. J. Membr. Biol. 80, 81–89
Nichols, D.G. (1982) Bioenergetics: an introduction to the chemiosmotic theory. Academic Press, London
Pope, A.J., Leigh, R.A. (1987) Some characteristics of anion transport at the tonoplast of oat roots, determined from the effects of anions on pyrophosphate-dependent proton transport. Planta 172, 91–100
Pope, A.J., Leigh, R.A. (1988) The use of a chloride-sensitive fluorescent probe to measure chloride transport in isolated tonoplast vesicles. Planta 176, 451–460
Pope, A.J., Jennings, I.R., Sanders, D., Leigh, R.A. (1990) Characterisation of Cl− transport in vacuolar membrane vesicles using a chloride-sensitive fluorescent probe. Reaction kinetic models for voltage- and concentration-dependence of Cl− flux. J. Membr. Biol. (in press)
Rea, P.A., Poole, R.J. (1985) Proton-translocating inorganic pyrophosphatase in red beet (Beta vulgaris L.) tonoplast vesicles. Plant Physiol. 77, 46–52
Rea, P.A., Leigh, R.A., Sanders, D. (1987) Wash activated Na +/ H+ exchange in tonoplast vesicles from Beta vulgaris storage root disks. In: Plant vacuoles: their importance in solute compartmentation and their applications in plant biotechnology, pp. 227–233, Marin, B., ed. Plenum Press, New York
Rudel, R., Senges, J. (1972) Experimental myotonia in mammalian skeletal muscle: change in membrane properties. Pflugers Arch. 331, 324–334
Schroeder, J.I., Hedrich, R. (1989) Involvement of ion channels and active transport in osmoregulation and signalling of higher plant cells. Trends Biochem. Sci. 14, 187–192
Schumaker, K.H., Sze, H. (1987) Decrease in pH gradients in tonoplast vesicles by Cl− and NO -3 : evidence for H+-coupled anion transport. Plant Physiol. 83, 490–496
Sze, H. (1985) H+-translocating ATPases: advances using membrane vesicles. Annu. Rev. Plant Physiol. 36, 175–208
Ticku, M.K., Olsen, R.W. (1977) γ-aminobutyric acid-stimulated chloride permeability in crayfish muscle. Biochim. Biophys. Acta 464, 519–529
Tyerman, S.D., Findlay, G.P. (1989) Current-voltage curves of single Cl− channels which coexist with two types of K+ channel in the tonoplast of Chara corallina. J. Exp. Bot. 40, 105–117
Tyerman, S.D., Findlay, G.P., Paterson, G.J. (1986) Inward membrane current in Chara inflata. II. Effects of pH, Cl− channel blockers and NH +4 , and significance of the hyperpolarised state. J. Membr. Biol. 89, 153–161
Welsh, M.J. (1983) Inhibition of chloride secretion by furosemide on canine tracheal epithelium. J. Membr. Biol. 71, 219–226
Welsh, M.J. (1984) Anthracene-9-carboxylic acid inhibits an apical membrane chloride conductance in canine tracheal epithelium. J. Membr. Biol. 78, 61–71
Wolfbeis, O.S., Urbano, E. (1982) Synthesis of fluorescent dyes. XIV. Standards for fluorescence measurements in the near-neutral pH range. J. Heterocyclic Chem. 19, 841–843
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pope, A.J., Leigh, R.A. Characterisation of chloride transport at the tonoplast of higher plants using a chloride-sensitive fluorescent probe. Planta 181, 406–413 (1990). https://doi.org/10.1007/BF00195895
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00195895