Abstract
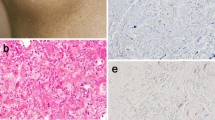
The expression and distribution of tenascin, an extracellular matrix glycoprotein, was investigated immunohistochemically using an anti-human tenascin monoclonal antibody (RCB 1) in formalin-fixed paraffin-embedded tissues obtained from 79 patients with skin appendage tumours, and compared with adjacent normal skin. Tissue specimens were pretreated with actinase and processed by the labelled streptavidin-biotin method. In normal skin, tenascin immunoreactivity was consistently found around the ductal portion of the sweat glands, around the lower part of the hair follicle and hair bulbs, and around or within blood vessels. Immunoreactivity was also observed variably around secretory coils of the sweat glands, and below the epidermis. No immunoreactivity was seen around the sebaceous glands. Tumours originating from sweat glands and hair follicles expressed tenascin around the tumour cells nests, while sebaceous gland tumours were immunonegative. Thus, tenascin expression in skin appendage tumours generally resembled that in corresponding normal tissue.
Similar content being viewed by others
References
Anbazhagan R, Sakakura T, Gusterson BA (1990) The distribution of immuno-reactive tenascin in the epithelial-mesenchymal junctional areas of benign and malignant squamous epithelia. Virchows Arch [B] 59: 59–63
Chiquet M, Fambrough DM (1984) Chick myotendinous antigen. I. A monoclonal antibody as a marker for tendon and muscle morphogenesis. J Cell Biol 98: 1926–1936
Chiquet-Ehrismann R, Mackie EJ, Pearson CA, Sakakura T (1986) Tenascin: an extracellular matrix protein involved in tissue interactions during fetal development and oncogenesis. Cell 47: 131–139
Crossin KL, Hoffman S, Grumet M, Thiery JP, Edelman GM (1986) Site-restricted expression of cytotactin during development of the chicken embryo. J Cell Biol 102: 1917–1930
Hashimoto K, Mehregan AH, Kumakiri M (1987) Tumors of skin appendages. (Practical dermatopathology series). Butterworths, Boston
Kallioinen M, Autio-Harmainen H, Dammert K, Risteli J, Risteli L (1984) Basement membrane laminin and type IV collagen in various benign and malignant adnexal tumors of the skin: an immunoperoxidase study. J Invest Dermatol 83: 276–280
Koukoulis GK, Gould VE, Bhattacharyya A, Gould JE, Howeedy AA, Virtanen I (1991) Tenascin in normal, reactive, hyperplastic, and neoplastic tissues: biologic and pathologic implications. Hum Pathol 22: 636–643
Lightner VA, Gumkowski F, Bigner DD, Erickson HP (1989) Tenascin/hexabrachion in human skin: biochemical identification and localization by light and electron microscopy. J Cell Biol 108: 2483–2493
Mackie EJ, Thesleff I, Chiquet-Ehrismann R (1987) Tenascin is associated with chondrogenic and osteogenic differentiation in vivo and promotes chondrogenesis in vitro. J Cell Biol 105: 2569–2579
Oike Y, Hiraiwa H, Kawakatsu H, Nishikai M, Okinaka T, Suzuki T, Okada A, Yatani R, Sakakura T (1990) Isolation and characterization of human fibroblast tenascin: an extracellular matrix glycoprotein of interest for developmental studies. Int J Dev Biol 34: 309–317
Sakakura T, Ishihara A, Yatani R (1991) Tenascin in mammary gland development: from embryogenesis to carcinogenesis. In: Lippmann M, Dickson R (eds) Regulatory mechanism in breast cancer. Kluwer Academic, Boston, pp 383–400
Schalkwijk J, Van Vlijmen I, Oosterling B, Perret C, Koopman R, Van den Born J, Mackie J (1991) Tenascin expression in hyperproliferative skin diseases. Br J Dermatol 124: 13–20
Shoji T, Kamiya T, Tsubura A, Hatano T, Sakakura T, Yamamoto M, Morii S (1992) Immunohistochemical staining patterns of tenascin in invasive breast carcinomas. Virchows Arch [A] 421: 53–56
Stamp GWH (1989) Tenascin distribution in basal cell carcinomas. J Pathol 159: 225–229
Tsubura A, Morii S, Ueda S, Sasaki M, Zotter St, Watzig V, Mooi W, Hagemen PC, Hilkens J, Van den Tweel J, Meijer C, Hilgers J (1987) Immunohistochemical demonstration of MAM-3 and MAM-6 antigens in normal human skin appendages and their tumors. Arch Dermatol Res 279: 550–557
Tsubura A, Okada H, Sasaki M, Dairkee SH, Morii S (1991) Immunohistochemical demonstration of keratins 8 and 14 in benign tumours of the skin appendage. Virchows Arch [A] 418: 503–507
Tsubura A, Fujita Y, Sasaki M, Morii S (1992) Lectin-binding profiles for normal skin appendages and their tumors. J Cutan Pathol 19: 483–489
Tsubura A, Senzaki H, Sasaki M, Hilgers J, Morii S (1992) Immunohistochemical demonstration of breast-derived and/or carcinoma associated glycoproteins in normal skin appendages and their tumors. J Cutan Pathol 19: 73–79
Wick MR, Swanson PE (1991) Cutaneous adnexal tumors. A guide to pathologic diagnosis. ASCP Press, Chicago
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shikata, N., Oyaizu, T., Tsubura, A. et al. Tenascin expression in normal human adult skin and skin appendage tumours. Vichows Archiv A Pathol Anat 424, 511–516 (1994). https://doi.org/10.1007/BF00191437
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00191437