Summary
-
1.
Larval cultures of Sarcophaga argyrostoma, “preconditioned” for a low incidence of pupal diapause by embryonic exposure to continuous light, were transferred from sequences of diapause-inducing long nights into terminal sequences of either diapause-averting short nights or darkness. Conversely, other groups preconditioned for a high incidence of diapause by embryonic exposure to long nights were transferred from short nights into terminal long nights or darkness.
-
2.
The inductive effects of long nights (diapause) and short nights (non-diapause) were accumulated during the larval sensitive period which continued until puparium formation. Since the larvae in any one group formed puparia over a number of days it follows that late pupariaters experienced a greater number of inductive light cycles (or days in DD) than those that pupariated early.
-
3.
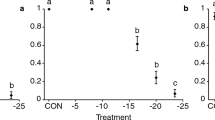
Analysis of diapause incidence in cultures transferred from long nights to DD at different temperatures, as a function of the day of pupariation, showed that the daily increase in diapause was a temperature-compensated process beginning after 9–10 cycles and reaching 10% after about 15 cycles. Cultures transferred from a series of short nights into DD at different temperature showed no evidence of temperature-compensation.
-
4.
The results are interpreted in terms of the external coincidence model for photoperiodic time measurement.
Similar content being viewed by others
References
Bradley HK, Saunders DS (1985) The selection for early and late pupariation in the flesh-fly Sarcophaga argyrostoma, and its effect on the incidence of pupal diapause. Physiol Entomol 10:371–382
Bünning E (1936) Die endogene Tagesrhythmik als Grundlage der photoperiodischen Reaktion. Ber Dt Bot Ges 54:590–607
Denlinger DL (1971) Embryonic determination of pupal diapause in the flesh fly Sarcophaga crassipalpis. J Insect Physiol 17:1815–1822
Fraenkel G, Hsiao C (1968) Manifestations of a pupal diapause in two species of flies, Sarcophaga argyrostoma and S. bullata. J Insect Physiol 14:689–705
Lewis RD, Saunders DS (1987) A damped circadian oscillator model of an insect photoperiodic clock. 1. Description of the model based on a feedback control system. J Theoret Biol 128:47–59
Pittendrigh CS (1966) The circadian oscillation in Drosophila pseudoobscura pupae: a model for the photoperiodic clock. Z Pflanzenphysiol 54:275–307
Pittendrigh CS (1972) Circadian surfaces and the diversity of possible roles of circadian organization in photoperiodic induction. Proc Natl Acad Sci USA 69:2734–2737
Pittendrigh CS (1981) Circadian organization and the photoperiodic phenomena. In: Follett BK, Follett DE (eds) Biological clocks in seasonal reproductive cycles. John Wright & Sons, Bristol, pp 1–35
Pittendrigh CS, Elliott J, Takamura T (1984) The circadian component in photoperiodic induction. In: Photoperiodic regulation of insect and molluscan hormones. Ciba Foundation Symposium 104:26–47
Saunders DS (1971) The temperature-compensated photoperiodic clock “programming” development and pupal diapause. J Insect Physiol 17:801–812
Saunders DS (1978a) An experimental and theoretical analysis of photoperiodic induction in the flesh-fly Sarcophaga argyrostoma. J Comp Physiol 124: 75–95
Saunders DS (1978b) Internal and external coincidence and the apparent diversity of photoperiodic clocks in the insects. J Comp Physiol 127:197–207
Saunders DS (1980) Some effects of constant temperature and photoperiod on the diapause response of the flesh fly, Sarcophaga argyrostoma. Physiol Entomol 5:191–198
Saunders DS (1981a) Insect photoperiodism: entrainment within the circadian system as a basis for time measurement. In: Follett BK, Follett DE (eds) Biological clocks in seasonal reproductive cycles. John Wright & Sons, Bristol, pp 67–80
Saunders DS (1981b) Insect photoperiodism — the clock and the counter: a review. Physiol Entomol 6:99–116
Saunders DS (1984) Photoperiodic time measurement in Sarcophaga argyrostoma: an attempt to use daily temperature cycles to distinguish external from internal coincidence. J Comp Physiol A 154:789–794
Saunders DS, Lewis RD (1987a) A damped circadian oscillator model of an insect photoperiodic clock. II. Simulations of the shapes of the photoperiodic response curves. J Theoret Biol 128:61–71
Saunders DS, Lewis RD (1987b) A damped circadian oscillator model of an insect photoperiodic clock. III. Circadian and “hourglass” responses. J Theoret Biol 128:73–85
Saunders DS, Lewis RD (1988) The photoperiodic clock and counter mechanism in two species of flies: evidence for damped circadian oscillators in time measurement. J Comp Physiol A 163:365–371
Vaz Nunes M, Kenny NAP, Saunders DS (1990) The photoperiodic clock in the blowfly Calliphora vicina. J Insect Physiol 36:61–67
Vaz Nunes M, Lewis RD, Saunders DS (1991a) A coupled oscillator feedback system as a model for the photoperiodic clock in insects and mites. I. The basic control system as a model for circadian rhythms. J Theoret Biol (in press)
Vaz Nunes M, Saunders DS, Lewis RD (1991b) A coupled oscillator feedback system as a model for the photoperiodic clock in insects and mites. II. Simulations of photoperiodic responses. J Theoret Biol (in press)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Saunders, D.S. The photoperiodic clock and “counter” in Sarcophaga argyrostoma: experimental evidence consistent with “external coincidence” in insect photoperiodism. J Comp Physiol A 170, 121–127 (1992). https://doi.org/10.1007/BF00190406
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00190406