Abstract
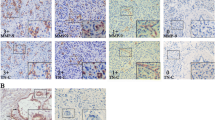
Wie studied the expression of tenascin (Tn) in human stomach. In the normal mucosa of the antrum and body, Tn reaction was only seen in the muscularis mucosae, in the region of the pyloric sphincter and in the duodenum, a Tn-immunoreactive rim was seen underlying surface epithelial cells. Antral gastritis, irrespective of the degree of inflammation, showed a rim-like Tn expression under the surface epithelial cells but no Tn reaction was seen in mild chronic gastritis of the body. In some moderate and severe examples of chronic gastritis a delicate Tn-reactive line was seen to underline the surface epithelium focally and the neck regions of gastric pits. Discontinuous Tn immunoreactivity was sometimes seen beneath hyperplastic epithelium in both parts of the stomach. A Tn-immunoreactive line was seldom seen surrounding glands showing intestinal metaplasia. In both benign and malignant ulcers prominent Tn immunoreaction was seen at the base of ulcers extending deep into the underlying muscularis. Only severely dysplastic lesions displayed Tn in the lamina propria, in close association with capillaries. In early forms of diffuse gastric cancer (DGCA) raggedly increased Tn staining was seen in the lamina propria underlying affected surface epithelial cells. In advanced forms of DGCA consistent Tn expression was seen in the tumour stroma. A distinct Tn reaction was seen surrounding invasive tumour cell nests of intestinal type gastric cancer (IGCA) in the submucosa, whereas in early forms of the tumour enhanced Tn reaction was noted predominantly in the upper part of the lamina propria in the vicinity of dysplastic elements. Notably, while most invading DGCA tumour cell nests showed no Tn in the submucosa and muscle cell layer, invading IGCA islets showed prominent expression of Tn. The most conspicuous Tn enhancement in the stomach is seen in invasive tumours and in ulcers suggesting that Tn is an important stromal component in malignant growth and in lesions undergoing active repair and remodelling.
Similar content being viewed by others
References
Chiquet-Ehrismann R (1990) What distinguishes tenascin from fibronectin? FASEB J 4:2598–2604
Chiquet-Ehrismann R, Mackie EJ, Pearson CA, Sakakura T (1986) Tenascin: an extracellular matrix protein involved in tissue interactions during fetal development and oncogenesis. Cell 47:131–139
Chuong CM, Chen HM (1991) Enhanced expression of neural cell adhesion molecules and tenascin (cytotactin) during wound healing. Am J Pathol 138:427–440
Eastwood GL (1977) Progress in gastroenterology. Gastrointestinal epithelial renewal. Gastroenterology 72:962–976
Ekblom P, Aufderheide E (1989) Stimulation of tenascin expression in mesenchyme by epithelial-mesenchymal interactions. Int J Dev Biol 33:7179
Erickson HP (1989) Tenascin: an extracellular matrix protein prominent in specialized embryonic tissues and tumors. Annu Rev Cell Biol 5:71–92
Gould VE, Martinez-Lacabe V, Virtanen I, Sahlin KM, Schwartz MM (1992) Differential distribution of tenascin and cellular fibronectins in acute and chronic renal allograft rejection. Lab Invest 67:71–79
Howeedy AA, Virtanen I, Laitinen L, Gould NS, Koukoulis GK, Gould VE (1990) Differentiat distribution of tenascin in the normal, hyperplastic, and neoplastic breast. Lab Invest 63:798–806
Jones FS, Burgoon MP, Hoffman S, Crossin KL, Cunningham BA, Edelman GM (1988) A cDNA clone for cytotactin contains sequences similar to epidermal growth factor-like repeats and segments of fibronectin and fibrinogen. Proc Natl Acad Sci USA 85:2186–2190
Kehrl JH, Wakefield LM, Roberts AB, Jakowlew S, Alvarez-Noon M, Derynck R, Sporn MB, Fauci AS (1986) Production of transforming growth factor β by human T lymphocytes and its potential role in the regulation of T cell growth. J Exp Med 163:1037–1050
Kekki M, Sipponen P, Siurala M, Laszewicz W (1990) Peptic ulcer and chronic gastritis: their relation to age and sex, and to location of ulcer and gastritis. Gastroenterol Clin Biol 14:217–223
Koukoulis GK, Gould VE, Bhattacharyya A, Gould JE, Howeedy AA, Virtanen I (1991) Tenascin in normal, reactive, hyperplastic and neoplastic tissues. Human Pathol 22:636–643
Lauren P (1965) The two histological main types of gastric carcinoma diffuse and so-called intestinal type carcinoma. Acta Pathol Microbiol Scand 64:31–49
Luomanen M, Virtanen I (1993) Distribution of tenascin in healing incision, excision and laser wounds. J Oral Pathol Med 22:41–45
Mackie EJ, Halfter W, Liverani D (1998) Induction of tenascin in healing wounds. J Cell Biol 107:2757–2767
Natali PG, Nicotra MR, Bigotti A, Botti C, Castellani P, Risso Am, Zardi L (1991) Comparative analysis of the expression of the extracellular matrix protein tenascin in normal human fetal, adult and tumor tissues. Int J Cancer 47:811–816
Pearson CA, Pearson D, Shibahara S, Hofsteenge J, Chiquet-Ehrismann R (1988) Tenascin: cDNA cloning and induction by TGF-β. EMBO J 7:2977–2981
Probstmeier R, Martini R, Schachner M (1990) Expression of J1/tenascin in the crypt-villus unit of adult mouse small intestine: implications for its role in epithelial cell shedding. Development 109:313–321
Riedl SE, Faissner A, Schlag P, Herbay A von, Koretz K, Möller P (1992) Altered content and distribution of tenascin in colitis, colon adenoma, and colorectal carcinoma. Gastroenterology 103:400–406
Sakai T, Kawakatsu H, Hirota N, Yokoyama T, Sakakura T, Saito M (1993) Specific expression of tenascin in human colonic neoplasms. Br J Cancer 67:1058–1064
Schalkwijk J, Steijlen PM, Vlijmen-Willems IMJJ van, Oosterling B, Mackie EJ, Verstraeten AA (1991) Tenascin expression in human dermis is related to epidermal proliferation. Am J Pathol 139:1143–1150
Shoji T, Kamiya T, Tsubura A, Hatano T, Sakakura T, Yamamoto M, Morii S (1992) Immunohistochemical staining patterns of tenascin in invasive breast carcinomas. Virchows Arch [A] 421:53–56
Sipponen P, Kekki M, Siurala M (1991) The Sidney system: epidemiology and natural history of chronic gastritis. J Gastroenterol Hepatol 6:244–251
Siri A, Carnemolla B, Saginati M, Leprini A, Casari G, Baralle F, Zardi L (1991) Human tenascin: primary structure, pre-mRNA splicing patterns and localization of the epitopes recognized by two monoclonal antibodies. Nucleic Acids Res 19:525–531
Tiitta O, Wahlström T, Paavonen J, Linnala A, Sharma S, Gould VE, Virtanen I (1992) Enhanced tenascin expression in cervical and vulvar koilocytotic lesions. Am J Pathol 141:907–913
Tiitta O, Wahlström T, Virtanen I, Gould VE (1993) Tenascin in inflammatory conditions and neoplasms of the urinary bladder. Virchows Arch [B] 63:283–287
Todd R, Donoff BR, Chiang T, Chou MY, Elovic A, Gallagher GT, Wong DTW (1991) The eosinophil as a cellular source of transforming growth factor alpha in healing cutaneous wounds. Am J Pathol 138:1307–1313
Vollmer G, Siegal GP, Chiquet-Ehrismann R, Lightner VA, Arnholdt H, Knuppen R (1990) Tenascin expression in the human endometrium and in endometrial adenocarcinomas. Lab Invest 62:725–730
Wright PA, Quirke P, Attanoos R, Williams GR (1992) Molecular pathology of gastric carcinoma: Progress and prospects. Hum Pathol 23:848–869
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tiitta, O., Virtanen, I., Sipponen, P. et al. Tenascin expression in inflammatory, dysplastic and neoplastic lesions of the human stomach. Vichows Archiv A Pathol Anat 425, 369–374 (1994). https://doi.org/10.1007/BF00189574
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00189574