Abstract
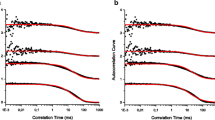
The subnanosecond fluorescence and motional dynamics of the tryptophan residue in the bacteriophage M13 coat protein incorporated within pure dioleoylphosphatidylcholine (DOPC) as well as dioleoylphosphatidylcholine/dioleoylphosphatidylglycerol (DOPC/DOPG) and dimyristoylphosphatidylcholine/dimyristoylphosphatidylglycerol (DMPC/DMPG) bilayers (80/20 w/w) with various L/P ratios have been investigated. The fluorescence decay is decomposed into four components with lifetimes of about 0.5, 2.0, 4.5 and 10.0 ns, respectively. In pure DOPC and DOPC/DOPG lipid bilayers, above the phase transition temperature, the rotational diffusion of the protein molecules contributes to the depolarization and the anisotropy of tryptophan is fitted to a dual exponential function. The longer correlation time, describing the rotational diffusion of the whole protein, shortens with increasing temperature and decreasing protein aggregation number. In DMPC/DMPG lipid bilayers, below the phase transition, the rotational diffusion of the protein is slowed down such that the subnanosecond anisotropy decay of tryptophan in this system reflects only the segmental motion of the tryptophan residue. Because of a heterogeneous microenvironment, the anisotropy decay must be described by three exponentials with a constant term, containing a negative coefficient and a negative decay time constant. From such a decay, the tryptophan residue within the aggregate undergoes a more restricted motion than the one exposed to the lipids. At 20°C, the order parameter of the transition moment of the isolated tryptophan is about 0.9 and that for the exposed one is about 0.5.
Similar content being viewed by others
Abbreviations
- DOPC:
-
1,2-dioleoyl-sn-glycero-3-phosphocholine
- DOPG:
-
1,2-dioleoyl-sn-glycero-3-phosphoglycerol
- DMPC:
-
1,2-dimyristoyl-sn-glycero-3-phosphocholine
- DMPG:
-
1,2-dimyristoylsn-glycero-3-phosphoglycerol
- L/P ratio:
-
phospholipid to coat protein molar ratio
- <τ>:
-
average fluorescence lifetime
- r (0):
-
initial anisotropy
- r (∝):
-
residual anisotropy
- S :
-
order parameter of the transition moment of the tryptophan residue
References
Alcala JR, Gratton E, Prendergast FG (1987a) Fluorescence lifetime distributions in proteins. Biophys J 51:597–604
Alcala JR, Gratton E, Prendergast FG (1987b) Interpretation of fluorescence decays in proteins using continuous lifetime distributions. Biophys J 51:925–936
Ameloot M, Hendrickx H, Herreman W, Pottel H, van Cauwelaert F, van der Meer W (1984) Effect of orientational order on the decay of the fluorescence anisotropy in membrane suspensions. Experimental verification on unilamellar vesicles and lipid/ α-lactalbumin complexes. Biophys J 46:525–539
Andrews LJ, Forster LS (1974) Fluorescence characteristics of indoles in non-polar solvents: lifetimes, quantum yields and polarization spectra. Photochem Photobiol 19:353–360
Beechem JM, Brand L (1985) Time-resolved fluorescence of proteins. Ann Rev Biochem 54:43–71
Chang MC, Petrich JW McDonald DB, Fleming GR (1983) Nonexponential fluorescence decay of tryptophan, tryptophylglycine and glycyltryptophan. J Am Chem Soc 105:3819–3824
Chapman D, Williams RM, Ladbrooke BD (1967) Physical studies of phospholipids. VI. Thermotropic and lyotropic mesomorphism of some 1,2-diacylphosphatidylcholines (lecithins). Chem Phys Lipids 1:445–475
Chen LX-Q, Longworth JW, Fleming GR (1987) Picosecond timeresolved fluorescence of ribonuclease T1. A pH and substrate analogue binding study. Biophys J 51:865–873
Cherry RJ (1979) Rotational and lateral diffusion of membrane proteins. Biochim Biophys Acta 559:289–327
Cherry RJ, Godfrey RE (1981) Anisotropic rotation of bacteriorhodopsin in lipid membranes. Comparison of theory with experiment. Biophys J 36:257–276
Cherry RJ, Müller U, Henderson R, Heyn MP (1978) Temperaturedependent aggregation of bacteriorhodopsin in dipalmitoyl- and dimyristoylphosphatidylcholine vesicles. J Mol Biol 121:283–298
Creed D (1984) The photophysics and photochemistry of the nearUV absorbing amino acids I. Tryptophan and its simple derivatives. Photochem Photobiol 39:537–562
Cross AJ, Fleming GR (1984) Analysis of time resolved fluorescence anisotropy decays. Biophys J 46:45–56
Cross AJ, Waldeck DH, Fleming GR (1983) Time-resolved polarization spectroscopy: level kinetics and rotational diffusion. J Chem Phys 78:6455–6467
Crowell RL, Lonberg-Holm K (1986) Virus attachment and entry into cell. Am Soc Microbiol, Washington DC, USA
Datema KP, Visser AJWG, van Hoek A, Wolfs CJAM, Spruijt RB, Hemminga MA (1987a) Time-resolved tryptophan fluorescence anisotropy investigation of bacteriophage M13 coat protein in micelles and mixed bilayers. Biochemistry 26:6145–6152
Datema KP, Wolfs CJAM, Marsh D, Watts A, Hemminga MA (1987b) Spin label electron spin resonance study of bacteriophage M13 coat protein incorporation into mixed lipid bilayers. Biochemistry 26:7571–7574
Datema KP, Van Boxtel BJH, Hemminga MA (1988) Dynamic properties of M13 coat protein in mixed bilayers. A deuterium NMR study of exchangeable proton sites. J Magn Reson 77:372–376
Devaux PF, Seigneuret M (1985) Specificity of lipid-protein interaction as determined by spectroscopic techniques. Biochim Biophys Acta 822:63–125
Heyn MP (1979) Determination of lipid order parameters and rotational correlation times from fluorescence depolarization experiments. FEBS Lett 108:359–364
Hochstrasser RM, Negus DK (1984) Picosecond fluorescence decay of tryptophans in myoglobin. Proc Natl Acad Sci USA 81:4399–4403
Hudson B, Harris DL, Ludescher RD, Ruggiero A, Cooney-Freed A, Cavalier SA (1986) In: Taylor DL, Waggoner AS, Lanni F, Murphy RF, Birge R (eds) Fluorescence in the biological sciences. Liss, New York
Jähnig F (1979) Structural order of lipids and proteins in membranes: Evaluation of fluorescence anisotropy data. Proc Natl Acad Sci USA 76:6361–6365
Johnson ID, Hudson BS (1989) Environmental modulation of M13 coat protein tryptophan fluorescence dynamics. Biochemistry 28:6392–6400
Kawato S, Kinosita K Jr (1981) Time-dependent absorption anisotropy and rotational diffusion of proteins in membranes. Biophys J 36:277–296
Kinosita K Jr, Kawato S, Ikegami A (1977) A theory of fluorescence polarization decay in membranes. Biophys J 20:289–305
Kinosita K Jr, Ikegami A, Kawato S (1982) On the wobbling in cone analysis of fluorescence anisotropy decay. Biophys J 37:461–464
Knippers R, Hoffmann-Berling H (1966) A coat protein from bacteriophage fd I. Interaction of the protein with DNA in vitro. J Mol Biol 21:281–292
Lakowicz JR, Maliwal BP, Cherek H, Balter A (1983) Rotational freedom of tryptophan residues in proteins and peptides. Biochemistry 22:1741–1752
Lipari G, Szabo A (1980) Effect of vibrational motion on fluorescence depolarization and nuclear magnetic resonance relaxation in macromolecules and membranes. Biophys J 30:489–506
Löfroth JE (1985) Deconvolution of single photon counting data with a reference convolution method and global analysis. Eur Biophys J 13:45–58
Ludescher RD, Volwerk JJ, de Haas GH, Hudson BS (1985) Complex photophysics of the single tryptophan of porcine pancreatic phospholipase A2, its zymogen, and enzyme/micelle complex. Biochemistry 24:7240–7249
Ludescher RD, Peting L, Hudson S, Hudson B (1987) Time-resolved fluorescence anisotropy for systems with lifetime and dynamic heterogeneity. Biophys Chem 28:59–75
Lumry R, Hershberger M (1978) Status of indole photochemistry with special reference to biological applications. Photochem Photobiol 27:819–840
Marvin DA, Wachtel EJ (1975) Structure and assembly of filamentous bacterial viruses. Nature 253:19–23
Munro I, Pecht I, Stryer L (1979) Subnanosecond motions of tryptophan residues. Proc Natl Acad Sci USA 76:56–60
Nakashima Y, Kongisberg W (1974) Reinvestigation of a region of the fd bacteriophage coat protein sequence. J Mol Biol 88:598–600
Nozaki Y, Chamberlain BK, Webster RE, Tanford C (1976) Evidence for a major conformational change of coat protein in assembly of fl bacteriophage. Nature 259:335–337
O'Connor DV, Phillips D (1984) Time-correlated single photon counting. Academic Press, London
Petrich JW Chang MC, McDonald DB, Fleming GR (1983) On the origin of nonexponential fluorescence decay in tryptophan and its derivatives. J Am Chem Soc 105:3824–3832
Ross JBA, Rousslang KW, Brand L (1981a) Time-resolved fluorescence and anisotropy decay of the tryptophan in adrenocorticotropin-(1–24). Biochemistry 20:4361–4369
Ross JBA, Schmidt CJ, Brand L (1981b) Time-resolved fluorescence of the two tryptophans in horse liver alcohol dehydrogenase. Biochemistry 20:4369–4377
Spatz L, Strittmatter P (1971) A form of cytochrome b5 that contains an additional hydrophobic sequence of 40 amino acid residues. Proc Natl Acad Sci USA 68:1042–1046
Szmacinski H, Jayaweera R, Cherek H, Lakowicz JR (1987) Demonstration of an associated anisotropy decay by frequency-domain fluorometry. Biophys Chem 27:233–241
Tanaka F, Mataga N (1982) Dynamic depolarization of interacting fluorophores. Effect of internal rotation and energy transfer. Biophys J 39:129–140
Tanaka F, Mataga N (1987) Fluorescence quenching dynamics of tryptophan in proteins. Effect of internal rotation under potential barrier. Biophys J 51:487–495
Valeur B, Weber G (1977) Resolution of the fluorescence excitation spectrum of indole into the 1 L a and 1 L b excitation bands. Photochem Photobiol 25:441–444
Van der Meer W, Pottel H, Herreman W, Ameloot M, Hendrickx H, Schröder H (1984) Effect of orientational order on the decay of the fluorescence anisotropy in membrane suspensions. A new approximate solution of the rotational diffusion equation. Biophys J 46:515–523
Van Hoek A, Vervoort J, Visser AJWG (1983) A subnanosecond resolving spectrofluorimeter for analysis of protein fluorescence kinetics. J Biochem Biophys Methods 7:243–254
Van Hoek A, Vos K, Visser AJWG (1987) Ultrasensitive time-resolved polarized fluorescence spectroscopy as a tool in biology and medicine. IEEE J Quantum Electron QE-23:1812–1820
Vaz WLC, Austin RH, Vogel H (1979) The rotational diffusion of cytochrome b 5 in lipid bilayer membranes. Biophys J 26:415–426
Visser AJWG, van Hoek A (1979) The measurement of subnanosecond fluorescence decay of flavins using time-correlated photon counting and a mode-locked Ar ion laser. J Biochem Biophys Methods 1:195–208
Visser AJWG, Vos K, van Hoek A, Santema JS (1988) Time-resolved fluorescence depolarization of rhodamine B and octadecyl rhodamine B in triton X-100 micelles and aerosol OT reversed micelles. J Phys Chem 92:759–765
Vos K, van Hoek A, Visser AJWG (1987) Application of a reference convolution method to tryptophan fluorescence in proteins. A refined description of rotational dynamics. Eur J Biochem 165:55–63
Wickner W (1975) Asymmetric orientation of a phage coat protein in cytoplasmic membrane of Escherichia coli. Proc Natl Acad Sci USA 72:4749–4753
Wickner W (1976) Asymmetric orientation of phage M13 coat protein in Escherichia coli cytoplasmic membranes and in synthetic lipid vesicles. Proc Natl Acad Sci USA 73:1159–1163
Wilson ML, Dahlquist FW (1985) Membrane protein conformational change dependent on the hydrophobic environment. Biochemistry 24:1920–1928
Wolber PK, Hudson BS (1982) Bilayer acyl chain dynamics and lipid-protein interaction. The effect of the M13 bacteriophage coat protein on the decay of the fluorescence anisotropy of parinaric acid. Biophys J 37:253–262
Zannoni C, Arcioni A, Cavatorta P (1983) Fluorescence depolarization in liquid crystals and membrane bilayers. Chem Phys Lipids 32:179–250
Zidovetzki R, Yarden Y, Schlessinger J, Jovin TM (1981) Rotational diffusion of epidermal growth factor complexed to cell surface receptors reflects rapid microaggregation and endocytosis of occupied receptors. Proc Natl Acad Sci USA 78:6981–6985
Author information
Authors and Affiliations
Additional information
On leave of Shanghai Medical Equipment Research Institute, 77 Jiang Ning Rd. Shanghai, People's Republic of China
Offprint requests to: M. A. Hemminga
Rights and permissions
About this article
Cite this article
Peng, K., Visser, A.J.W.G., van Hoek, A. et al. Analysis of time-resolved fluorescence anisotropy in lipid-protein systems. Eur Biophys J 18, 285–293 (1990). https://doi.org/10.1007/BF00188041
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00188041