Abstract
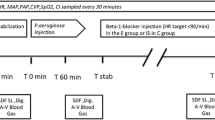
The epithelial damage and the accumulation of the leucocytes within intestinal wall layers after ischemia and reperfusion was investigated in a pig model. Superior mesenteric artery (SMA) was occluded for 1 h (group 2, n = 9), 2 h (group 3, n = 6) and 3 h (group 4, n = 7) with a consecutive 2 h reperfusion period. The histological evaluation was performed on hematoxylin-eosine and Naphtol AS-D chloracetate stained preparations.
The intensity of reperfusion shock depended on the duration of the intestinal ischemia. After 1 h SMA occlusion systolic blood pressure stabilized at a lower level with a normalization of the serum lactate level and the intestinal intramural pHi within the reperfusion period. After 2 h SMA occlusion the decrease of the systolic blood pressure was intensified (54–69 mm Hg) with a persistent elevated serum lactate concentration and a delayed increase of the ischemic pHi values. Reperfusion after 3 h SMA occlusion caused an irreversible shock.
The epithelial damage also depended on the duration of the SMA occlusion. There were no significant changes of the leucocytic accumulation within the submucosa. But a significant increase of the number of the leucocytes was seen within the inner and the outer layer of the muscularis after 1 h SMA occlusion (106±5/mm2 resp. 280/mm2; p<0.05). This increase was less pronounced after 2 h (92±5/mm2 *resp. 189±4/mm2; *p<0.05) and 3 h of SMA occlusion (84±5/mm2 resp. 185±23/mm2).
Intestinal ischemia and reperfusion caused no changes of the leucocytic accumulation within the submucosa but a significantly increased accumulation within the muscularis after 1 h SMA occlusion, which was not seen after a more elongated occlusion period. A reperfusion shock without normalization of the serum lactate level and the intramural pHi suggesting intestinal perfusion disturbances may also lead to a depression of the leucocytic accumulation within the muscularis.
Zusammenfassung
Der Epithelschaden und die Leukozytenakkumulation in der Wand des Ileums nach Ischämie und Reperfusion wurden experimentell am Schwein untersucht. Die A. mesenterica superior wurde für 1 h (Gruppe 2; n=9), 2 h (Gruppe 3; n=6) und 3 h (Gruppe 4; n=7) abgeklemmt und 2 h reperfundiert. Die histologische Beurteilung erfolgte an Hämatoxylin-Eosin- und NaphtolAS-D-Chlorazetatesterase-gefärbten Präparaten. Es entwickelte sich ein Reperfusionsschock in Abhängigkeit von der Ischämiedauer. Nach lstündiger Ischämie stabilisierte sich der Blutdruck mit erniedrigten Werten sowie einer Normalisierung des Serumlaktatspiegels und des intramuralen pHi des Dünndarms. Eine Verlängerung der Ischämie auf 2 h (Gruppe 3) führte zu einem Blutdruckabfall (54–69 mmHg), einem bleibend erhöhten Serumlaktatspiegel (5,2–6,0 mmol/1) und einem verzögerten Anstieg des pHi. Die Reperfusion nach 3stündiger Ischämie verursachte einen irreversiblen Schock. Der Epithelschaden des Dünndarms war abhängig von der Ischämiedauer. Es fand sich, kein Unterschied der Leukozytenakkumulation innerhalb der Submukosa im Vergleich zur Kontrollgruppe. Ein signifikanter Anstieg der Leukozytenzahl war jedoch in der Lamina interna und externa der Muskularis nach lstündiger Ischämie zu verzeichnen (106±5/mm2 bzw. 280/mm2; p<0,05), der nach 2stündiger [92±5/mm2 (p<0,05) bzw. 189±4/mm2 und 3stündiger Ischämiedauer (84±5/mm2 bzw. 185±23/mm2) nur geringgradig erhöht war. Während die intestinale Ischämie und der anschließende reperfusionsbedingte Schock nicht zu einer Beeinflussung der Leukozytenakkumulation innerhalb der Submukosa, aber zu einer signifikanten Akkumulation innerhalb der Muskularis nach einer lstündigen Ischämie führten, scheint bei ausbleibender Normalisierung des Serumlaktatspiegels und einem verzögerten oder fehlenden Anstieg der ischämischen intramuralen pHi-Werte als Hinweis auf eine intestinale Perfusionsstörung als Folge einer längeren Ischeämiedauer die Akkumulation der Leukozyten innerhalb der Muscularis mucosae abgescwächt zu sein.
Similar content being viewed by others
Literatur
Amano H, Bulkley GB, Gorey T, Hamilton SR, Horn SD, Zuidema GD (1980) Role of the microvascular patency of small intestine from ischemic injury. Surg Forum 31:157–159
Antonsson JB, Boyle CC, Kruithoff KL, Wang H, Sacristan E, Rothschild HR, Fink PM (1990) Validation of tonometric measurement of gut intramural pH during endotoxemia and mesenteric occlusion in pigs. Am J Physiol 259:G519-G523
Barroso-Aranda J, Schmid-Schoenbein GW, Zweifach BW, Engler RL (1988) Granulocytes and no-reflow phenomenon in irreversible hemorrhagic shock. Circ Res 63:437–447
Boyd AJ, Sherman IA, Saibi FG (1994) Intestinal microcirculation and leukocyte behavior in ischemia-reperfusion injury. Microvasc Res 47:355–368
Bulkley GB, Kvietys PR, Parks DA, Perry MA, Granger DN (1986) Relationship of blood flow and oxygen consumption to ischemic injury in the canine small intestine. Gastroenterology 89:852–857
Chiu CJ, McArdle AH, Brown R, Scott HJ, Gurd FN (1979) Intestinal mucosal lesion in low flow states. Arch Surg 101: 478–483
Chin CJ, Scott HJ, Gurd FN (1972) Volume deficit versus toxic absorption: a study of canine shock after mesenteric arterial occlusion. Ann Surg 175:479–488
Fiddian-Green RG (1989) Studies in splanchnic ischemia and multiple organ failure. In: Marston A, Bulkley GB, FiddianGreen RG, Haglund U (eds) Splanchnic ischemia and multiple organ failure. Arnold, London und Mosby, St Louis, pp 349–363
Fiddian-Green RG, Haglund U, Gitierrez G, Shoemaker WC (1993) Goals for the resuscitation of shock. Crit Care Med 21:S25-S31
Granger DN, Hollwarth ME, Parks DA (1986) Ischemia/reperfusion injury: role of oxygen-derived free radicals. Acta Physiol Scand 548:47–53
Granger DN, Benoit JN, Suzuki M, Grisham MB (1989) Leucocyte adherence to venular endothelium during ischemia-reperfusion. Am J Physiol 257:G704-G708
Grisham MB, Hernandez LA, Granger DN (1986) Xanthine oxidase and the neutrophil infiltration in the intestinal ischemia. Am J Physiol 251:G567-G574
Grum CM, Fiddian-Green RG, Pittenger GL, Grant BJB, Rothman ED, Dantzker DR (1984) Adequacy of tissue oxygenation in intact dog intestine. J Appl Physiol 56:1065–1069
Grum CM (1993) Tissue oxygenation in low flow states and during hypoxemia. Crit Care Med 21:S44-S49
Haglund U, Bulkley GB, Granger DN (1987) On the pathophysiology of the intestinal ischemic injury. Eur J Surg 153:321–324
Haglund U (1993) Systemic mediators released from the gut. Crit Care Med 21:S15-S18
Hansell P, Borgstrom P, Arfors KE (1993) Pressure related capillary leukostasis following ischemia-reperfusion and hemorrhagic shock. Am J Physiol 265:H381-H388
Horton J, White DJ (1991) Cardiac contractile injury after intestinal ischemia-reperfusion. Am J Physiol 261:H1164-HI1170
Jamieson WG,De Rose G, Harris KA, Pliaging G, Stafford L (1993) Myocardial and circulating performance during the ischemic phase of superior mesenteric artery occlusion. Can J Surg 36:435–439
Jonas J, Schwarz S, Alebrahim-Dehkordy A (1995) Das Verhalten des Laktatspiegels bei Okklusion and Reperfusion der A. mesenterica superior. Langenbecks Arch im Druck
Kubes P, Suzuki M, Granger DN (1990) Platelet activating factor-induced microvascular dysfunction: role of adherent leukocytes. Am J Physiol 258:G158-G163
Kurose I, Anderson DC, Miyasaka M, Tamatani T, Paulson JC, Todd RF, Rusche JR, Granger DN (1994) Molecular determinants of reperfusion-induced leukocyte adhesion and vascular protein leakage. Circ Res 74:336–343
Kurtel H, Tso P, Granger DN (1992) Granulocyte accumulation in postischemic intestine: role of the leukocyte glycoprotein CD 11/CD 18. Am J Physiol 262:G878-G882
Mazzoni MC, Borgstrom P, Intaglietta M, Arfors K (1990) Capillary narrowing in hemorrhagic shock is rectified by hyperosmotic saline-dextran reinfusion. Circ Shock 31:407–418
Moloney WC, McPheson L, Fliegelman L (1960) Esterase activity in leucocytes demonstrated by the use of naphtol AS-D chlorazetate substrate. J Histochem Cytochem 200–207
Nelson DP, King CE, Dodd SL, Schmacker PT, Cain SM (1987) Systemic and intestinal limits of O2 extraction in the dog. J Appl Physiol 63:387–394
O'Neill P, Cobb LM, Steigmann CK, Chaudry IH (1993) Prevention of secondary cardiovascular instability after intestinal ischemia and reperfusion improves survival. Am J Physiol 264:R622-R629
Park PO, Haglund U, Bulkley GB, Fdlt K (1990) The sequence of development of intestinal tissue injury after strangulation ischemia and reperfusion. Surgery 107:574–580
Parks DA, Shah AK, Granger DN (1984) Oxygen radicals: effects on intestinal vacular permeability. Am J Physiol 247:G167-G170
Parks DA, Granger DN (1986) Contributions of ischemia and reperfusion to mucosal lesion formation. Am J Physiol 250: G749-G753
Perry MA, Granger DN (1992) Leukocyte adhesion in local versus hemorrhage-induced ischemia. Am J Physiol 263:H810-H815
Reilly P, Bulkley GB (1993) Vasoactive mediators and the splanchnic circulation. Crit Care Med 21:S55-S68
Strecker U, Dick W, Madjidi A, Ant M (1993) The effect of the type of colloid on the efficacy of hypertonic saline colloid mixtures in hemorrhagic shock. Resuscitation 25:41–57
Tjiong B, Bella E, Weiner M, Enquist IF (1974) Fluid shifts and metabolic changes during and after occlusion of the superior mesenteric artery. Surg Gynecol Obstet 139:217–221
Welbourn CRB, Goldman G, Kobzik L, Paterson I, Valeri CR, Shepro D, Hechtman HB (1990) Neutrophil adherence receptors (CD 18) in ischemia. J Immunol 145:1906–1911
Welbourn CRB, Goldman G, Paterson IS, Valeri CR, Shepro D, Hechtman HB (1991) Pathophysiology of ischemia reperfusion injury: central role of the neutrophil. Br J Surg 78:651–655
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jonas, J., Heimann, A., Alebrahim-Dehkordy, A. et al. Reperfusionsschock nach okklusion der A. mesenterica superior und akkumulation von leukozyten innerhalb der dünndarmwand. Langenbecks Arch Chir 381, 95–101 (1996). https://doi.org/10.1007/BF00183939
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00183939