Summary
Flow cytometry (FCM) has been used to evaluate not only the malignancy of tumor cells but also the effects of chemotherapy. Here a new application of FCM for selecting the best antineoplastic agent in the chemotherapy for brain tumors is reported.
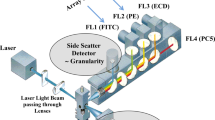
Through our preliminary study using established brain tumor cell lines, the system for this sensitivity test was developed. Antineoplastic agents were placed in contact with monolayer-cultured cells; then cell viability and changes in the DNA histogram were analyzed by FCM. Cell viabilities were measured with the fluorescein diacetate (FDA) staining method, and the DNA histogram was analyzed by the propidium iodide (PI) staining method. The best antineoplastic agent was determined based on changes in cell viability and cell cycle. In other words, when markedly decreased viability as compared with that of the control, is measured by FCM, then the agents can be considered to have had a cytocydal effect on the tumor cells, and thus the sensitivity of the agents is able to be evaluated. If the viability of the tumor cell is observed to be similar to that of the control, the cytostatic effects of the agents are able to be evaluated only if a marked change is observed in the DNA histogram.
After the preliminary study, this system was applied clinically to malignant brain tumor cases, resulting in success in selecting the best antineoplastic agent for each individual case. Our sensitivity test using this FCM established in vitro system has much potential value for clinical use.
Similar content being viewed by others
References
Kawamoto K, Herz F, Wolley RC, Hirano A, Kajikawa H, Koss LG: Flow cytometric analysis of the DNA distribution in human brain tumors. Acta Neuropath 46:39–44, 1979
Kawamoto K, Nishiyama T, Ikeda Y, Yamanouchi Y, Kawamura Y, Matsumura H, Hirano A, Herz F, Wolley RC: Flow cytometric studies of human brain tumors — Part I: Human malignant brain tumors. Neurol Surg 8:723–728, 1980
Kawamoto K, Herz F, Wolley RC, Hirano A, Koss LG: Flow cytometric analysis of the DNA content in cultured human brain tumor cells. Virchows Arch B Cell Path 35:11–17, 1980
Kawakami K, Kawamoto K, Oka N, Kawamura Y, Matsumura H, Ito T, Ohyama A: Flow cytometric studies on brain tumors — Part 5: New sensitivity test of antineoplastic agents for brain tumors and its clinical application. Neurol Surg 14:627–634, 1986
Pullen GR, Chalmers PJ, Nind APP, Nairn RC: Criteria of cell killing in vitro. J Immunol Methods 43:87–93, 1981
Oka N, Kawamoto K, Kawakami K, Fujiwara H, Shoda Y, Matsumura H: Experimental study on measurement of viability using fluorescein diacetate. Kansai Flow Cytometry 2:58–64, 1985 (in Japanese)
Vindelov LL: Flow microfluorometric analysis of nuclear DNA in cells from solid tumors and cell suspensions. Virchows Arch B Cell Path 24:227–242, 1977
Yoshida J, Cravioto H, Ransohoff J: In vitro transformation of fetal brain cells from CDF rats exposed in utero to N-Ethyl-N-nitrosourea: Morphological and immunologic studies. JNCI 64:1231–1239, 1980
Kawakami K, Kawamura Y, Kawamoto K, Oka N, Numa Y, Matsumura H, Akagi K, Ohyama A: Multidisciplinary therapy for gliomas. Experimental study using brain tumor-bearing rats and analytical study of clinical cases. Neurol Med Chir (Tokyo) 26:133–139, 1986
Taguchi T: History and significance of sensitivity test of anticancer drugs. Cancer Chemother 9:570–574, 1982
Bickis IJ, Henderson IWD, Quastel JH: Biochemical studies of human tumors. II. In vitro estimation of individual tumor sensitivity to anticancer agents. Cancer 19:103–113, 1966
Salmon SE, Hamburger AW, Soehnlen B, Durie BGM, Alberts DS, Moon TE: Quantitation of differential sensitivity of human-tumor stem cells to anticancer drugs. New Engl J Med 298:1321–1327, 1978
Hamburger AW, Salmon SE: Primary bioassay of human tumor stem cells. Science 197:461–463, 1977
von Hoff DD, Casper J, Bradley E, Sandbach J, Jones D, Makuch R: Association between human tumor colony-forming assay results and response of an individual patient's tumor to chemotherapy. Am J Med 70:1027–1032, 1981
Inoue M, Arakawa M, Ogawa K: Human tumor stem cell assay. Cancer Chemother 9:599–605, 1982
Nishiyama T, Kawamura Y, Kawamoto K, Matsumura H, Ito T, Ohyama A: Cultivation of human brain tumor cells on the chorioallantoic membrane of fertile chicken eggs. Neuropathology 3:13–28, 1982
Kornblith PL, Szypko PE: Variations in response of human brain tumors to BCNU in vitro. J Neurosurg 48:580–586, 1978
Shibuya N, Yoshida J, Kobayashi T, Kageyama N: Antitumor activity of ACNU against malignant glioma — A clinical application of in vitro sensitivity test of chemotherapeutic agent. Neurol Surg 11:361–367, 1983
Rosenblum ML, Dougherty DV, Deen DF, Wilson CB: Potentials and limitations of a clonogenic cell assay for human brain tumors. Cancer Treat Rep 65 (Suppl 2):61–66, 1981
Shapiro WR, Basler GA, Chernik NL, Posner JB: Human brain tumor transplantation into nude mice. J Natl Cancer Inst 62:447–453, 1979
Takamoto S, Ota K: Mechanism of action of antitumor agents by flow microfluorometry. Cancer Chemother 5:727–736, 1978
Shitara N, Kohno T, Nagamune A, Takakura K, Sano K: Pulsecytophotometric studies on experimental brain tumor under the effect of chemotherapeutic agents, microwave irradiation and hyperthermia. Neurol Med Chir (Tokyo) 18:199–207, 1978
Nomura K, Hoshino T, Knebel K, Deen DF, Barker M: BCNU-induced perturbations in the cell cycle of 9L rat brain tumor. Cancer Treat Rep 62:747–754, 1978
Ford WL: The preparation and labelling of lymphocytes. In DM Weir (ed) Handbook of Experimental Immunology, 3rd ed, Blackwell Scientific, Oxford, 1978, pp Chap 23, 1–22
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kawamoto, K., Kawakami, K., Kawamura, Y. et al. New sensitivity test using flow cytometry. J Neuro-Oncol 6, 361–370 (1988). https://doi.org/10.1007/BF00177434
Issue Date:
DOI: https://doi.org/10.1007/BF00177434