Abstract
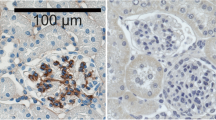
Cell-matrix interactions play a fundamental role in normal and pathological conditions. They can be mediated by the cytoadhesin subgroup of the integrin superfamily of adhesion molecules. Its members include the vitronectin receptor (VNR) and the platelet glycoprotein IIb/IIIa (GP IIb/IIIa). Both receptors are composed of an α-chain (αv and aIIb, respectively) coupled to a β3-chain. Using in situ immunohistochemistry and monoclonal antibodies, the authors studied the distribution of GP IIIa (common β3-chain), GP IIb/IIIa (αIIb-chain) and VNR (αv-chain) in normal and pathological corneal tissues. In the normal cornea, the limbal vascular endothelium was weakly αv-positive. Occasionally, faint and granular staining was seen in the epithelium. In the pathological samples, an upregulated expression of the αv chain was noticed on the corneal epithelium as well as on fibroblasts and corneal endothelium. The aIIb and β3-chains were consistently absent. These data suggest that expression of the VNR-αv- chain in the human cornea is modulated by soluble factors released during inflammation and wound healing. Dissociation of expression of the αv and β3-chains suggests usage of an alternative α-chain by the VNR-αv-chain.
Similar content being viewed by others
References
Albelda SM, Buck CA (1990) Integrins and other cell adhesion molecules. FASEB J 4:2868–2880
Athanasou NA, Quinn J, Horton MA, McGee J'OD (1990) New sites of normal vitronectin receptor immunoreactivity detected with osteoclast-reacting monoclonal antibodies 13C2 and 2306. Bone Miner 8:7–22
Bodary SC, McLean JW (1990) The integrin β1 subunit associates with the vitronectin receptor αv subunit to form a novel vitronectin receptor in a human embryonic kidney cell line. J Biol Chem 265:5938–5941
Buck CA, Horwitz AF (1987) Cell surface receptors for extracellular matrix molecules. Ann Rev Cell 13:179–205
Charo IF, Bekaert LS, Phillips DR (1987) Platelet glycoprotein IIb/IIIa-like proteins mediate endothelial cell attachment to adhesive protein and extracellular matrix. J Biol Chem 262:9935–9938
Charo IF, Nannizzi L, Smith JW, Cheresh DA (1990) The vitronectin receptor αvβ3 binds fibronectin and acts in concert with α5β1 in promoting cellular attachment and spreading on fibronectin. J Cell Biol 111:2795–2800
Cheng YF, Kramer RH (1989) Human microvascular endothelial cells express integrin-related complexes that mediate adhesion to the extracellular matrix. J Cell Physiol 139:275–286
Cheresh DA (1987) Human endothelial cells synthesize and express an arg-gly-asp-directed adhesion receptor involved in attachment to fibrinogen and van Willebrand factor. Proc Natl Acad Sci USA 84:6471–6475
Cheresh DA, Spiro RC (1987) Biosynthetic and functional properties of an Arg-Gly-Asp directed receptor involved in human melanoma cell attachment to vitronectin, fibrinogen, and von Willebrand factor. J Biol Chem 262:17703–17711
Cheresh DA, Smith JW, Cooper HM, Quaranta V (1989) A novel vitronectin receptor integrin (αvβx) is responsible for distinct adhesive properties of carcinoma cells. Cell 57:56–69
Dedhar S, Gray V (1990) Isolation of a novel integrin receptor mediating Arg-Gly-Asp-directed cell adhesion to fibronectin and type I collagen from human neuroblastoma cells. Association of a novel β1-related subunit with αv. J Cell Biol 110:2185–2193
Dejana E, Colella S, Conforti G, Abbadini M, Gaboli M, Marchisio PC (1988) Fibronectin and vitronectin regulate the organization of their respective Arg-Gly-Asp adhesion receptors in cultured human endothelial cells. J Cell Biol 107:1215–1223
Dejana E, Languino LR, Colella S, Corbascio GC, Plow E, Ginsberg M, Marchisio PC (1988) The localization of a platelet glycoprotein IIb/IIIa-related protein in endothelial cell adhesion structures. Blood 71:566–572
Fitzgerald LA, Poncz M, Steiner B, Rall SC, Benneth JS, Phillips DR (1987) Comparison of cDNA-derived protein sequences of the human Fibronectin and vitronectin receptor α-subunits and platelet glycoprotein IIb. Biochemistry 26:8158–8165
Fitzgerald LA, Steiner B, Rall SC, Lo SS, Phillips DR (1987) Protein sequence of endothelial glycoprotein IIIa derived from a cDNA clone. J Biol Chem 262:3936–3939
Freed E, Gailit J, Geer P van der, Ruoslahti E, Hunter T (1989) A novel integrin β subunit is associated with the vitronectin receptor α subunit (αv) in a human osteosarcoma cell line and is substrate for protein kinase C. EMBO J 8:2955–2965
Ginsberg MH, Loftus JC, Plow EF (1988) Cytoadhesins, integrins and platelets. Thromb Haemost 59:1–6
Hayashi K, Frangeih G, Wolf G, Kenyon KR (1989) Expression of transgrowing growth factor-β in wound healing of vitamin A-deficient rat corneas. Invest Ophthalmol Vis Sci 30:239–247
Hayman EG, Pierschbacher MD, Öhgren Y, Ruoslahti E (1983) Serum spreading factor (vitronectin) is present at the cell surface and in tissues. Proc Nail Acad Sci USA 80:4003–4007
Hemler ME (1990) VLA-proteins in the Integrin family: structures, functions and their role on leucocytes. Ann Rev Immunol 8:365–400
Hynes RO (1987) Integrins: a family of cell surface receptors. Cell 48:549–554
Ignatius MJ, Large TH, Houde M, Tawil JW, Burton A, Esch F, Carbonetto S, Reichardt LF (1990) Molecular cloning of the rat integrin α1-subunit: a receptor for laminin and collagen. J Cell Biol 111:709–720
Ignotz RA, Heino J, Massagué J (1989) Regulation of cell adhesion receptors by transgrowing growth factor-β: regulation of vitronectin receptor and LFA-1. J Biol Chem 264:389–392
Kay EP (1986) Rabbit corneal endothelial cells modulated by polymorphonuclear leucocytes are fibroblasts: comparison with keratocytes. Invest Ophthalmol Vis Sci 27:891–897
Kishimoto TK, Larson RS, Corbi AL, Dustin ML, Staunton DE, Springer TA (1989) The leucocyte integrins. Adv Immunol 46:149–182
Kramer RH, Cheng YF, Clyman R (1990) Human microvascular endothelial cells use β1 and β3, Integrin receptor complexes to attach to laminin. J Cell Biol 111:1233–1243
Lauweryns B, van den Oord JJ, Volpes R, Foets B, Missotten L (1991) Distribution of very late activation integrins in the human cornea: an immunohistochemical study using monoclonal antibodies. Invest Ophthalmol Vis Sci 32:2079–2085
Lawler J, Weinstein R, Hynes RO (1988) Cell attachment to thrombospondin: role of Arg-Gly-Asp, calcium and integrin receptors. J Cell Biol 107:2351–2361
Nazzarro V, Berti E, Cerri A, Brusasco A, Cavalli R, Caputo R (1990) Expression of integrins in junctional and dystrophic epidermolysis bullosa. J Invest Dermatol 95:60–64
Phillips DR, Charo IF, Parise LV, Fitzgerald LA (1988) The platelet membrane glycoprotein IIb/IIIa complex. Blood 71:831–843
Podack ER, Preissner KT, Müller Eberhard HJ (1984) Inhibition of C9 polymerisation within the SC5b-9 complex of complement by S-protein. Acta Pathol Microbiol Immunol Scand (C) 92S:889–896
Podack ER, Dahlbäck B, Griffin J (1986) Interaction of S-protein of complement with thrombin and antithrombin III during coagulation. J Biol Chem 261:7387–7392
Postlewhaite AE, Keski-Oja J, Moses HL, Kang AH (1987) Stimulation of the chemotactic migration of human fibroblasts by transgrowing growth factor beta. J Exp Med 165:251–256
Pytela R, Pierschbacher MD, Ruoslahti E (1985) A 125/115 kD cell surface receptor specific for vitronectin interacts with the arginine-glycine-aspartic acid adhesion sequence derived from fibronectin. Proc Natl Acad Sci USA 82:5766–5770
Pytela R, Pierschbacher MD, Ginsberg MH, Plow EF, Ruoslahti E (1986) Platelet membrane glycoprotein IIb/IIIa: member of a family of Arg-Gly-Asp-specific adhesion receptors. Science 231:1559–1562
Rapraeger A, Bernfield M (1985) Cell surface proteoglycan of mouse mammary epithelial cells: protease releases a heparan sulfate-rich ectodomain from a putative membrane-anchored domain. J Biol Chem 260:4103–4109
Roberts AB, Sporn MB, Assoain RK, Smith JM, Roche NS, Wakefield LM, Heine UI, Liotta LA, Falanga V, Kehrl JH, Fauci AS (1986) Transgrowing growth factor type-beta: rapid induction of fibrosis and angiogenesis in vivo and stimulation of collagen formation in vitro. Proc Natl Acad Sci USA 83:4167–4171
Ruoslahti E, Pierschbacher MD (1987) New perspectives in cell adhesion: RGD and integrins. Science 238:491–497
Sheppard D, Rozzo C, Starr L, Quaranta V, Erie DJ, Pytela R (1990) Complete amino acid sequence of a novel integrin β subunit (β6) identified in epithelial cells using the polymerase chain reaction. J Biol Chem 265:11502–11507
Smith JW, Vestal DJ, Irwin SV, Burke TA, Cheresh DA (1990) Purification and functional characterization of integrin αvβ5. An adhesion receptor for vitronectin. J Biol Chem 265:11008–11013
Sporn MB, Roberts AB, Wakefield LM, Crombrugghe B de (1987) Some recent advances in the chemistry and biology of transgrowing growth factor-β. J Cell Biol 105:1039–1045
Stepp MA, Park CC, Spurr-Michaud C, Gipson I (1990) Identification of integrins in stationary and migrating rat corneal epithelia. ARVO Abstracts. Invest Ophthalmol Vis Sci 31:538
Suzuki S, Naitch Y (1990) Amino acid sequence of a novel integrin β4 subunit and primary expression of the mRNA in epithelial cells. EMBO J 9:757–763
Suzuki S, Argraves WS, Arai H, Languino LR, Pierschbacher MD, Ruoslahti E (1987) Amino acid sequence of the vitronectin receptor α subunit and comparative expression of adhesion receptor mRNAs. J Biol Chem 262:14080–14085
Suzuki S, Argraves WS, Pytela R, Arai H, Kruisius T, Pierschbacher MD, Ruoslahti E (1986) cDNA and amino acid sequences of the cell adhesion protein receptor recognising vitronectin reveal a transmembrane domain and homologies with other adhesion protein receptors. Proc Natl Acad Sci USA 83:8614–8618
Vogel BE, Tarone G, Giancotti FG, Gailit J, Ruoslahti E (1990) A novel fibronectin receptor with an unexpected subunit composition (αvβ1). J Biol Chem 265:5934–5937
Author information
Authors and Affiliations
Additional information
Offprint requests to: B. Lauweryns
Rights and permissions
About this article
Cite this article
Lauweryns, B., van den Oord, J.J. & Missotten, L. Localization of the cytoadhesin integrins in the human cornea. Graefe's Arch Clin Exp Ophthalmol 230, 264–268 (1992). https://doi.org/10.1007/BF00176302
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00176302