Summary
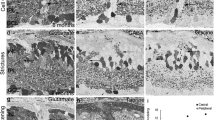
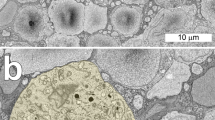
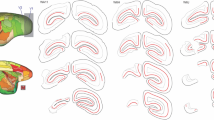
We have analysed the number and spatial distribution of displaced retinal ganglion cells in the frog Litoria (Hyla) moorei. A series of normal animals was compared with one in which the optic nerve was crushed and allowed to regenerate. Ganglion cells were labelled with horseradish peroxidase (HRP) applied to the optic nerve, and retinae were examined as sections or whole mounts. We analysed separately ganglion cells with somata displaced to the inner nuclear (Dogiel cells, DGCs) and to the inner plexiform layer (IPLGCs). These findings were related to data for the orthotopic ganglion cells (OGCs). The mean number of DGCs in the normal series was 2,550 (±281) and fell to 1,630 (±321) after regeneration, representing a mean loss of 36%. This reduction was not significantly different from the mean loss of 43% from the OGC population in which mean values fell from 474,700 (±47,136) to 268,700 (±54,395). In both the normal and the regenerate series, DGCs were estimated to represent means of only 0.6% of the OGC population. Densities of DGCs were highest in the nasoventral and temporo-dorsal peripheries; densities of both DGCs and OGCs were lower after optic nerve regeneration. We conclude that the factors which affect ganglion cell death during optic nerve regeneration, do so to similar extents amongst the DGC and the OGC populations. The IPLGCs were very rare in normal animals with a mean of 420 (±95). However, their numbers increased after regeneration to a mean of 3,350 (±690), estimated to be 1.2% of the OGC population. These cells normally favoured peripheral retina but became pan-retinal after regeneration. The primary dendrites of the majority of IPLGCs were oriented in the same direction as those of OGCs. We conclude that most IPLGCs were OGCs which had relocated their somata to the inner plexiform layer.
Similar content being viewed by others
References
Adams SC (1977) Technical considerations in the use of HRP as a neuronal marker. Neuroscience 2:141–146
Ball A, Dickson DH (1983) Displaced amacrine and ganglion cells in the newt retina. Exp Eye Res 36:199–213
Barker J, Grigg G (1977) A field guide to Australian frogs. Rigby, Perth Sydney
Beazley LD (1981) Retinal ganglion cell death and regeneration of abnormal retinotectal preojections after removal of a segment of optic nerve in Xenopus tadpoles. Dev Biol 85:164–170
Beazley LD, Darby JE, Perry VH (1986) Cell death in the retinal ganglion cell layer during optic nerve regeneration for the frog Rana pipiens. Vision Res 26:543–556
Beazley LD, Dunlop SA, Harman AM, Coleman L-A (1989) Development of cell density gradients in Amphibia and marsupials — two solutions to one problem. In: Finlay BL, Sengelaub D (eds) Development of the vertebrate retina. Plenum Press, pp 199–226
Boycott BB, Dowling JE (1969) Organisation of the primate retina: light microscopy. Philos Trans R Soc Lond [Biol] 255:109–184
Buhl EH, Dann JF (1988) Morphological diversity of displaced retinal ganglion cells: A Lucifer Yellow study. J Comp Neurol 269:210–218
Bunt AH, Minckler DS (1977) Displaced ganglion cells in the retina of monkey. Invest Ophthalmol Vis Sci 16:95–98
Coleman L-A, Dunlop SA, Beazley LD (1984) Patterns of cell division during visual streak formation in the frog Limnodynastes dorsalis. J Embryol Exp Morphol 83:119–135
Coleman L-A, Harman AM, Beazley LD (1987) Displaced retinal ganglion cells in the wallaby Setonix brachyurus. Vision Res 27:1269–1277
Darby JE, Carr RA, Beazley LD (1990) Retinal ganglion cell death during optic nerve regeneration is not accompanied by appreciable cell loss from the inner nuclear layer. Anat Embryol 182:487–492
Dogiel AS (1888) Ein besonderer Typus von Nervenzellen in der mittleren Gangliosen Schicht der Vogel-Retina. Anat Anz 3:133–143
Drager UC (1985) Birth dates of retinal ganglion cells giving rise to the crossed and uncrossed optic projections in the mouse. Proc R Soc Lond [Biol] 224:57–77
Dunlop SA, Humphrey MF, Beazley LD (1989) Displaced retinal ganglion cells in the frog Hyla moorei. Neurosci Lett [Suppl] 34:S78
Dunlop SA, Fraley SM, Beazley CD (in press) The morphology of developing and regenerating ganglion cells. In: Sharma SC, Fawcett J (eds) Formation and Regeneration of Nerve Connection, pp 42–53. Birkhäuser, Boston, MA
Ebbesson SOE, Tang D (1965) A method for estimating the number of cells in histological sections. J R Microsco Soc 84:449–464
Frank BD, Hollyfield JG (1987) Retinal ganglion cell morphology in the Frog, Rana pipiens. J Comp Neurol 266:413–434
Fite KV, Brecha N, Karten HJ, Hunt SP (1981) Displaced ganglion cells and the accessory optic system of the pigeon. J Comp Neurol 195:279–288
Gaze RM (1970) Formation of nerve connections. Academic Press, New York
Halasz P, Martin PR (1984) A microcomputer based system for semi-automatic analysis of histological sections. Proc R Microsc Soc 19:312P
Harman AM, Beazley LD (1989) Generation of retinal cells in the wallaby, Setonix brachyurus (quokka). Neuroscience 28:219–232
Holt CE, Bertsch TW, Ellis HM, Harris WA (1988) Cellular determination in the Xenopus retina is independent of lineage and birth date. Neuron 1:15–26
Humphrey MF (1985) The formation of nerve connections between the eye and the brain during optic nerve regeneration in the frog Hyla moorei. PhD Thesis, University of Western Australia
Humphrey MF (1987) Effect of different optic nerve lesions on retinal ganglion cell death in the frog Rana pipiens. J Comp Neurol 266:209–219
Humphrey MF (1988) A morphometric study of the retinal ganglion cell response to optic nerve severance in the frog Rana pipiens. J Neurocytol 17:293–304
Humphrey MF, Beazley LD (1985) Retinal ganglion cell death during optic nerve regeneration in the frog Hyla moorei. J Comp Neurol 236:382–402
Humphrey MF, Darby JE, Beazley LD (1989) Prevention of optic nerve regeneration in the frog Hyla moorei transiently delays the death of some ganglion cells. J Comp Neurol 279:187–198
Ito H, Murakami T (1984) Retinal ganglion cells in two teleost species, Sebastiscus marmoratus and Navodon modestus. J Comp Neurol 229:80–96
Linden R (1987) Displaced ganglion cells in the retina of the rat. J Comp Neurol 258:138–143
Liu Z-H, Jen LS (1986) Displaced retinal ganglion cells in normal rats and- rats with one eye enucleated at birth. Neurosci Lett 67:239–244
Montgomery N, Fite KV, Bengston L (1981) The accessory optic system of Rana pipiens. Neuroanatomical connections and intrinsic organization. J Comp Neurol 236:382–402
Perry VH (1981) Evidence for an amacrine cell system in the ganglion layer of the rat retina. Neuroscience 6:931–944
Reh TA (1987) Cell-specific regulation of neuronal production in the larval frog retina. J Neurosci 7:3317–3324
Ramon Y Cajal SR (1892) La retinae des vertibres. In La Cellule Vol 9:17–257. English translation: The structure of the retina (compiled and translated by Thorpe SA and Glickstein M) Thomas, Springfield, Ill
Rodieck RW (1979) Visual pathways. Ann Rev Neurosci 2:193–225
Scalia F, Arango V, Singman EL (1985) Loss and displacement of ganglion cells after optic nerve regeneration in adult Rana pipiens. Brain Res 344:267–280
Sheard PW, Beazley LD (1988) Retinal ganglion cell death is not prevented by application of tetrodotoxin during optic nerve regeneration in the frog Hyla moorei. Vision Res 28:461–470
Sperry RW (1951) Mechanisms of neural maturation. In: Stevens SS (ed) Handbook of experimental psychology. Wiley, New York, pp 236–280
Stelzner DJ, Strauss JA (1986) A quantitative analysis of frog optic nerve regeneration: is retrograde ganglion cell death or collateral axonal loss related to selective reinnervation? J Comp Neurol 245:83–106
Straznicky K, Gaze RM (1971) The growth of the retina in Xenopus laevis: a autoradiographic study. J Embryol Exp Morphol 26:67–79
Tachibana M (1978) Displaced ganglion cells in carp retina revealed by the horseradish peroxidase technique. Neurosci Lett 9:153–157
Tay D, Hiscock J, Straznicky C (1982) Temporo-nasal asymmetry in the accretion of retinal ganglion cells in late larval and postmetamorphic Xenopus. Anat Embryol 164:87–93
Tennant M, Masini LJ, Darby JE, Beazley LD (1992) Dimensions of the lesion site influence the extent of retinal ganglion cell death during optic nerve regeneration in the frog Littoria (Hyla) moorei. Proc Aust Neurosci Soc 3:97
Toth P, Straznicky C (1989) The morphological characterization and distribution of displaced ganglion cells in the Anuran retina. Visual Neurosci 3:551–561
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dunlop, S.A., Humphrey, M.F. & Beazley, L.D. Displaced retinal ganglion cells in normal frogs and those with regenerated optic nerves. Anat Embryol 185, 431–438 (1992). https://doi.org/10.1007/BF00174081
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00174081