Summary
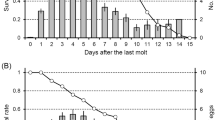
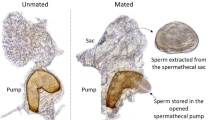
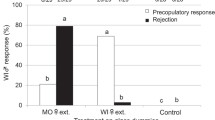
The primary sex ratio and the selective factors associated with it were investigated in the aphid Pemphigus spyrothecae. The sexuparae gave birth to an average of 1.99 ± 0.01 (1–2) males and 5.06 ± 0.09 (2–6) females [± SE (range), n = 147]. The average weight of individual males was 16.2±0.86 (13–18) μg (n = 5) and of females 40.7 ± 0.71 (38–46) μg (n = 15). The overall investment ratio by the mother sexuparae was therefore 1:6.4, equivalent to a proportionate investment in sons of 0.135 (interquartiel range: 0.117–0.165). There was a highly significant correlation between the size of the sexupara and the number of her daughters: the number of sons was independent of sexupara size. Field observations and laboratory experiments showed that the sexuparae aggregate on poplar bark before giving birth to the sexuals: they appear to behave in such a way that there is always at least one other sexupara in the crevice with them. The average number of sexuparae forming a “foundress” group, within which offspring could potentially mate with each other, was 2.48±0.19 (1–9) (n = 80). The life-time activity patterns of the sexuals (which moult four times but do not feed) was described. Both sexes are active shortly after birth; they are then immobile for a long period but become very active again after emerging as adults (after about 42 h in males, 51 h in females). Each male can mate with up to 14 females. The females are receptive to a second male for only a relatively short period (ca. 15–20 min) after their first mating. There is considerable competition between males during mating (just as intense between clonal sibs as non-sibs) and evidence for post-copulatory mate-guarding by the males. There is no evidence that females prefer to mate with non-sibs rather than sibs. The observed sex ratios and the mating behaviour of the aphids are discussed in relation to models of local mate competition assuming variable female fecundity, as developed by Frank (1987a, b) and Yamaguchi (1985) (the constant male hypothesis) and by Stubblefield and Seger (1990).
Similar content being viewed by others
References
Aoki S, Kurosu U, Stern DL (1991) Aphid soldiers discriminate between soldiers and non-soldiers, rather than between kin and non-kin, in Ceratoglyphina bambusae. Anim Behav 42:865–866
Blackman RL (1980) Chromosomes and parthenogenesis in aphids. In: Blackman RL, Hewitt GM, Ashburner M (eds) Insect cytogenetics. Symp R Entomol Soc Lond 10:133–148
Boomsma JJ, Isaaks JA (1985) Energy investment and respiration in queens and males of Lasius niger (Hymenoptera: Formicidae). Behav Ecol Sociobiol 18:19–27
Bull JJ, Charnov EL (1988) How fundamental are Fisherian sex ratios? In: Harvey PH, Partridge L (eds) Oxford surveys in evolutionary biology, vol 5. Oxford University Press, Oxford, pp 96–135
Clark PJ, Evans FC (1954) Distance to nearest neighbor as a measure of spatial relationships in population ecology. Ecology 35:445–453
Dixon AFG (1985) Aphid ecology. Blackie, Glasgow
Eastop VF, Hille Ris Lambers D (1976) Survey of the World's Aphids. Junk, The Hague
Eastop VF, van Emden HF (1972) The insect material. In: van Emden HF (ed) Aphid Technology. Academic Press, London New York, pp 1–45
Frank SA (1985) Hierarchical selection theory and sex ratios. II. On applying the theory, and a test with fig wasps. Evolution 39:949–964
Frank SA (1987a) Individual and population sex ratio allocation patterns. Theor Popul Biol 31:47–74
Frank SA (1987b) Variable sex ratios among colonies of ants. Behav Ecol Sociobiol 20:195–201
Green RF, Gordh G, Hawkins BA (1982) Precise sex ratios in highly inbred parasitic wasps. Am Nat 120:653–665
Hales DF, Mittler TE (1983) Precocene causes male determination in the aphid Myzus persicae. J Insect Physiol 29: 819–823
Hales DF, Mittler TE (1987) Endocrine control of male determination in aphids. In: Holman J, Pelikán J, Dixon AFG, Weismann L (eds) Population structure, genetics and taxonomy of aphids and Thysanoptera. SBP Academic Publishing, pp 158–164
Hales DF, Wellings PW, Parkes RA (1989) Investment in gynoparae and males by Myzus persicae (Sulzer). Funct Ecol 3:727–734
Hamilton WD (1967) Extraordinary sex ratios. Science 156:477–488
Heie OE (1980) The Aphidoidea (Hemiptera) of Fennoscandia and Denmark 1. Fauna Entomol Scand 9:1–236
Herre EA (1987) Optimality, plasticity and selective regime in fig wasp sex ratios. Nature 329:627–629
Kindlmann P, Dixon AFG (1989) Role of population density in determining the sex ratios of species that show local mate competition, with aphids as a model group. Funct Ecol 3:311–314
Kurosu U, Aoki S (1992) Why are aphid galls so rare? Evol Theor (in press)
Lampel G (1960) Die morphologischen und ökologischen Grundlagen des Generationswechsels monözischer und heterözischer Pemphiginen der Schwarz- und Pyramidenpappel. Z Angew Entomol 47: 334–375
Lampel G (1968–1969) Untersuchungen zur Morphologie von Pemphigus spirothecae Pass 1860 (Homoptera, Aphidoidea). Bull Naturforsch Gesellschaft Freiburg 58:56–72
May RM, Seger J (1985) Sex ratios in wasps and aphids. Nature 318:408–409
Newton C, Dixon AFG (1987) Cost of sex in aphids: size of males at birth and the primary sex ratio in Sitobion avenae (F.). Funct Ecol 1:321–326
Nunney L, Luck RF (1988) Factors influencing the optimum sex ratio in a structured population. Theor Popul Ecol 33:1–30
Orlando E (1974) Sex determination in Megoura viciae Buckton (Homoptera, Aphididae). Monit Zool Ital (N.S.) 8:61–70
Poole R (1974) Introduction to Quantitative Ecology. McGraw Hill, New York
Stroyan HLG (1964) Notes on some British species of Pemphigus Hartig (Homoptera: Aphidoidea) forming galls on Poplar, with the description of a new species. Proc R Entomol Soc Lond (B) 33:92–100
Stubblefield JW, Seger J (1990) Local mate competition with variable fecundity: dependence of offspring sex ratios on information utilization and mode of male production. Behav Ecol 1: 68–80
Werren JH (1980) Sex ratio adaptations to local mate competition in a parasitic wasp. Science 208:1157–1159
Yamaguchi Y (1985) Sex ratios of an aphid subject to local mate competition with variable maternal condition. Nature 318:460–462
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Foster, W.A., Benton, T.G. Sex ratio, local mate competition and mating behaviour in the aphid Pemphigus spyrothecae . Behav Ecol Sociobiol 30, 297–307 (1992). https://doi.org/10.1007/BF00170595
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00170595