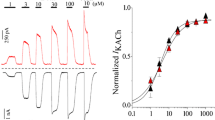
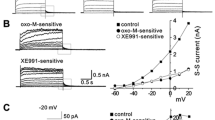
Abstract
We have compared muscarinic acetylcholine receptor (mAChR) coupling to phospholipase C (PLC) and increases in cytoplasmic Ca2+ concentration [Ca2+]i in human embryonic kidney (HEK) cells, stably expressing either the human m3 or m2 receptor subtype. In m3 mAChR-expressing cells, carbachol stimulated inositol phosphate (InsP) formation and increased [Ca2+]i with EC50 values of about 2 μM and 30 nM, respectively. Maximal inositol 1,4,5-trisphosphate (InsP3) production (about fourfold) was rapid (15 s) and stable for 2 min. Maximal increases in [Ca2+]i were 300–350 nM and mainly, almost 90%, due to influx of extracellular Ca2+. The efficacy of pilocarpine for stimulating InsP andCa2+ responses was not significantly different from that of carbachol. All m3 mAChR-mediated responses were pertussis toxin (PTX)-insensitive. In m2 mAChR-expressing cells, carbachol stimulated InsP formation and increased [Ca2+]i with EC50 values of about 20 μM and 7 μM, respectively. Maximal InsP formation was only 10–15% of that observed in m3 mAChR-expressing cells, whereas maximal elevations of [Ca2+]i were similar in both cell types. Formation of InsP3 was rapid (15 s to 2 min) and about twofold above basal. In contrast to m3 mAChR activation, [Ca2+]i increases induced by m2 mAChR activation were exclusively due to Ca2+ mobilization from intracellular stores.The efficacy of pilocarpine for stimulating InsP and Ca2+ responses was 50% and 20% of the efficacy of carbachol, respectively. PTX treatment did not affect m2 mAChR-induced PLC stimulation, but reduced the m2 mAChR-mediated increases in [Ca2+]i to 50%. In conclusion, m3 and m2 mAChRs stably expressed in HEK cells can induce similar cellular responses; however, they do so by activating apparently distinct signalling pathways. While coupling of m2 mAChR to PLC occurs in a PTX-insensitive manner, coupling to mobilization of Ca2+ from intracellular stores is partly PTX-sensitive and this may occur at least partly independent of PLC activation.
Similar content being viewed by others
References
Ashkenazi A, Peralta EG, Winslow JW, Ramachandran J, Capon DJ (1989) Functionally distinct G proteins selectively couple different receptors to PI hydrolysis in the same cell. Cell 56:487–493
Berridge MJ, Downes CP, Hanley MR (1982) Lithium amplifies agonist-dependent phosphatidylinositol responses in brain and salivary glands. Biochem J 206:587–595
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Clapham DE (1995) Calcium signaling. Cell 80:259–268
Dell'Acqua ML, Carroll RC, Peralta EG (1993) Transfected m2 muscarinic acetylcholine receptors couple to Gαi2 and Gαi2 in Chinese hamster ovary cells. Activation and desensitization of the PLC signaling pathway. J Biol Chem 268:5676–5685
Fasolato C, Innocenti B, Pozzan T (1994) Receptor-activated Ca2+ influx:how many mechanisms for how many channels? Trends Pharmacol Sci 15:77–83
Felder CC (1995) Muscarinic acetylcholine receptors: signal transduction through multiple effectors. FASEB J 9:619–625
Felder CC, Dieter P, Kinsella J, Tamura K, Kanterman RY, Axelrod J (1990) A transfected m5 muscarinic acetylcholine receptor stimulates phospholipase A2 by inducing both calcium influx and activation of protein kinase C. J Pharmacol Exp Ther 255: 1140–1147
Felder CC, Poulter MO, Wess J (1992) Muscarinic receptor-operated Ca2+ influx in transfected fibroblast cells is independent of inositol phosphates and release of intracellular Ca2+. Proc Natl Acad Sci USA 89:509–513
Feth F, Rascher W, Michel MC (1991) G-protein coupling and signalling of Y1-like neuropeptide Y receptors in SK-N-MC cells. Naunyn-Schmiedeberg's Arch Pharmacol 344:1–7
Frelin C, Brettmayer JP, Vigne P (1993) ADP induces inositol phosphate-independent intracellular Ca2+ mobilization in brain capillary endothelial cells. J Biol Chem 268:8787–8792
Galione A (1993) Cyclic ADP-ribose: a new way to control calcium. Science 259:325–326
Hosey MM (1992) Diversity of structure, signaling and regulation within the family of muscarinic cholinergic receptors. FASEB J 6:845–852
Irvine RF (1992) Inositol phosphates and Ca2+ entry: toward a proliferation or a simplification? FASEB J 6:3085–3091
Lechleiter J, Peralta E, Clapham D (1989) Diverse functions of muscarinic acetylcholine receptor subtypes. Subtypes of muscarinic receptors Trends Pharmacol Sci [Suppl] IV:34–38
Lustig KD, Conklin BR, Herzmark P, Taussig R, Bourne HR (1993) Type II adenylyl cyclase integrates coincident signals from Gs, Gi, and Gq, J Biol Chem 268:13900–13905
Michel MC, Brass L, Williams A, Bokoch GM, LaMorte VJ, Motulsky HJ (1989) α2-Adrenergic receptor stimulation mobilizes intracellular Ca2+ in human erythroleukemia cells. J Biol Chem 264:4986–4991
Michel MC, Feth F, Stieneker M, Rascher W (1992) NPY and carbachol raise Ca2+ in SK-N-MC cells by three different mechanisms. Naunyn-Schmiedeberg's Arch Pharmacol 345:370–374
Mihara S-I, Shigeri Y, Fujimoto M (1989) Neuropeptide Y-induced intracellular Ca2+ increases in vascular smooth muscle cells. FEBS Lett 259:79–82
Motulsky HJ, Michel MC (1988) Neuropeptide Y mobilizes Ca2+ and inhibits adenylate cyclase in human erythroleukemia cells. Am J Physiol 255:E880-E885
Neher E, Marry A, Fukuda K, Kuba T, Numa S (1988) Intracellular calcium release mediated by two muscarinic receptor subtypes. FEBS Lett 240:88–94
Offermanns S, Schultz G (1994) Complex information processing by transmembrane signaling systems involving G proteins. Naunyn-Schmiedeberg's Arch Pharmacol 350:329–338
Offermanns S, Wieland T, Homann D, Sandmann J, Bombien E, Spicher K, Schultz G, Jakobs KH (1994) Transfected muscarinic acetylcholine receptors selectively couple to Gi-type G proteins and Gq/11. Mol Pharmacol 45:890–898
Peralta EG, Ashkenazi A, Winslow JW, Ramachandran J, Capon DJ (1988) Differential regulation of PI hydrolysis and adenylyl cyclase by muscarinic receptor subtypes. Nature 334:434–437
Putney JW, Bird GSJ (1993) The signal for capacitative calcium entry. Cell 75:199–201
Sandmann J, Peralta EG, Wurtmann RJ (1991) Coupling of transfected muscarinic acetylcholine receptor subtypes to phospholipase D. J Biol Chem 266:6031–6034
Schmidt M, Hüwe SM, Fasselt B, Homann D, Rümenapp U, Sandmann J, Jakobs KH (1994) Mechanisms of phospholipase D stimulation by m3 muscarinic acetylcholine receptors. Evidence for involvement of tyrosine phosphorylation. Eur J Biochem 225:667–675
Smrcka AV, Sternweis PC (1993) Regulation of purified subtypes of phosphatidylinositol-specific phospholipase C\ by G protein α and \γ subunits. J Biol Chem 268:9667–9674
Tashjian AH Jr, Heslop JP, Berridge MJ (1987) Subsecond and second changes in inositol polyphosphates in GH4C1 cells induced by thyrotropin-releasing hormone. Biochem J0 243:305–308
Tobin AB, Lambert DG, Nahorski SR (1992) Rapid desensitization of muscarinic m3 receptor-stimulated polyphosphoinositide responses. Mol Pharmacol 42: 1042–1048
Willars GB, Nahorski SR (1995) Quantitative comparison of muscarinic and bradykinin receptor-mediated Ins(1,4,5)P3 accumulation and Ca2+ signalling in human neuroblastoma cells. Brit J Pharmacol 114:1133–1142
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schmidt, M., Bienek, C., van Koppen, C.J. et al. Differential calcium signalling by m2 and m3 muscarinic acetylcholine receptors in a single cell type. Naunyn-Schmiedeberg's Arch Pharmacol 352, 469–476 (1995). https://doi.org/10.1007/BF00169379
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00169379