Abstract
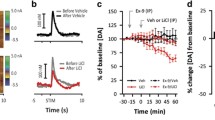
Fast cyclic voltammetry at carbon-fibre microelectrodes was used to investigate the effects of chronic clozapine or haloperidol administration on electrically evoked dopamine efflux in the nucleus accumbens and caudate putamen of the anaesthetized rat. Stimulation trains were delivered to the median forebrain bundle (60 pulses, 350 μs duration) every 5 min, and the evoked dopamine efflux measured as a function of a) the applied stimulus intensity (range 0.2 mA-1.0 mA), and b) the applied stimulus frequency (range 10 Hz-250 Hz). Chronic administration of either clozapine (20 mg/kg × 21 days, p.o.) or haloperidol (1 mg/kg × 21 days, p.o.) significantly reduced electrically evoked dopamine efflux in the nucleus accumbens over the range of stimulus intensities and frequencies tested. The reduction in evoked dopamine efflux observed in the nucleus accumbens of clozapine- and haloperidol-treated rats showed no statistically significant difference. In contrast, only chronic haloperidol treatment significantly reduced evoked dopamine efflux in the caudate putamen. These findings demonstrate that chronic treatment with either the atypical neuroleptic, clozapine, or the typical neuroleptic, haloperidol, produce long-term changes in mesolimbic dopamine function; actions which may underlie their antipsychotic efficacy. They also provide further evidence that the sparing action of clozapine on nigrostriatal dopamine activity may underlie the lower incidence of extrapyramidal side effects associated with its long-term administration.
Similar content being viewed by others
References
Armstrong-James M, Millar J (1979) Carbon fibre microelectrodes. J Neurosci Methods 1:279–287
Blaha CD, Lane RF (1987) Chronic treatment with classical and atypical antipsychotic drugs differentially decreases dopamine release in striatum and nucleus accumbens in vivo. Neurosci Lett 78:199–204
Bunney BS, Grace AA (1978) Acute and chronic haloperidol treatment: Comparison of effects on nigral dopaminergic cell activity. Life Sci 23:1715–1728
Chai B, Meltzer HY (1992) The effect of chronic clozapine on basal dopamine release and apomorphine-induced DA release in the striatum and nucleus accumbens as measured by in vivo brain microdialysis. Neurosci Lett 136:47–50
Chen J, Paredes W, Gardner EL (1991) Chronic treatment with clozapine selectively decreases basal dopamine release in nucleus accumbens but not in caudate-putamen as measured by in vivo brain microdialysis: further evidence for depolarization block. Neurosci Lett 122:127–131
Chesi AJR, Feasey-Truger KJ, Alzheimer C, ten Bruggencate G (1995) Dopamine autoreceptor sensitivity is unchanged in rat nucleus accumbens after chronic haloperidol treatment: an in vivo and in vitro voltammetric study. Eur J Neurosci 7:2450–2457
Chiodo LA, Bunney BS (1983) Typical and atypical neuroleptics: differential effects of chronic administration on the activity of A9 and A10 midbrain dopaminergic neurons. J Neurosci 3:1607–1619
Chiodo LA, Bunney BS (1985) Possible mechanisms by which repeated clozapine administration differentially affects the activity of two subpopulations of midbrain dopamine neurons. J Neurosci 5: 2539–2544
Farde L, Norström A-L, Wiesel F-A, Pauli S, Halldin C, Sedvall G (1992) Positron emmision tomographic analysis of central D1 and D2 receptor occupancy in patients treated with classical neuroleptics and clozapine. Relation to extrapyramidal side-effects. Arch Gen Psychiatry 49:538–544
Feasey-Truger KJ, Earl CD, Alzheimer C, ten Bruggencate G (1995a) Stimulus-evoked dopamine overflow in the rat nucleus accumbens is decreased following chronic haloperidol administration: an in vivo voltammetric study. Neurosci Lett 183:91–95
Feasey-Truger KJ, Alzheimer C, ten Bruggencate G (1995b) Chronic clozapine administration reduces stimulus-evoked dopamine overflow in the rat nucleus accumbens in vivo. Soc Neurosci. Abstr. 21, 1139
Grace AA, Bunney BS (1986) Induction of depolarization block in midbrain dopamine neurons by repeated administration of haloperidol: analysis using in vivo intracellular recording. J Pharmacol Exp Ther 238:1092–1100
Hernandez L, Hoebel BG (1989) Haloperidol given chronically decreases basal dopamine in the prefrontal cortex more than the striatum or nucleus accumbens as simultaneously measured by microdialysis. Brain Res Bull 22:763–769
Hernandez L, Hoebel BG (1995) Chronic clozapine selectively decreases prefrontal cortex dopamine as shown by simultaneous cortical, accumbens, and striatal microdialysis in freely moving rats. Pharmacol Biochem Behav 52:581–589
Ichikawa J, Meltzer HY (1990) Apomorphine does not reverse reduced nasal dopamine release in rat striatum and nucleus accumbens after chronic haloperidol treatment. Brain Res 507:138–142
Ichikawa J, Meltzer HY (1991) Differential effects of repeated treatment with haloperidol and clozapine on dopamine release and metabolism in the striatum and the nucleus accumbens. J Pharmacol Exp Ther 256:348–357
Invernizzi R, Morali F, Pozzi L, Samanin R (1990) Effects of acute and chronic clozapine on dopamine release and metabolism in the striatum and nucleus accumbens of conscious rats. Br J Pharmacol 100:744–748
Invernizzi R, Pozzi L, Samanin R (1995) Further studies on the effects of chronic clozapine on regional extracellular dopamine levels in the brain of conscious rats. Brain Res 670:165–168
Kuhr WG, Bigelow JC, Wightman RM (1986) In vivo comparison of the regulation of releasable dopamine in the caudate nucleus and the nucleus accumbens of the rat brain. J Neurosci 6:974–982
Lane RF, Blaha CD (1987) Chronic haloperidol decreases dopamine release in striatum and nucleus accumbens in vivo: depolarization block as a possible mechanism of action. Brain Res Bull 18:135–138
Lane RF, Blaha CD, Rivet JM (1988) Selective inhibition of mesolimbic dopamine release following chronic administration of clozapine: involvement of α1-noradrenergic receptors demonstrated by in vivo voltammerry. Brain Res 460:398–401
Laurelle M, Jaskiw GE, Lipska BK, Kolachana B, Casanova MF, Kleinman JE, Weinberger DR (1992) D1 and D2 receptor modulation in rat striatum and nucleus accumbens after subchronic and chronic haloperidol treatment. Brain Res 575:47–56
Mereu G, Lilliu V, Vargiu P, Muntoni AL, Diana M, Gessa GL (1994) Failure of chronic haloperidol to induce depolarization inactivation of dopamine neurons in unanesthetized rats. Eur J Pharmacol 264:449–453
Mereu G, Lilliu V, Vargiu P, Muntoni AL, Diana M, Gessa GL (1995) Depolarization inactivation of dopamine neurons: an artifact? J Neurosci 15:1144–1149
Millar J, Stamford JA, Kruk ZL, Wightman RM (1985) Electrochemical, pharmacological and electrophysiological evidence of rapid dopamine release and removal in the rat caudate nucleus following electrical stimulation of the median forebrain bundle. Eur J Pharmacol 109:341–348
Moghaddam B, Bunney BS (1993) Depolarization inactivation of dopamine neurons: terminal release characteristics. Synapse 14:195–200
Nowak JZ, Arbilla S, Galzin AM, Langer SZ (1983) Changes in sensitivity of release modulating dopamine autoreceptors after chronic treatment with haloperidol. J Parmacol Exp Ther 226: 558–564
O'Dell SJ, La Hoste GJ, Widmark CB, Shapiro RM, Potkin SG, Marshall JF (1990) Chronic treatment with clozapine or haloperidol differentially regulates doparnine and serotonin receptors in rat brain. Synapse 6:146–153
O'Donnell P, Grace AA (1995) Different effects of subchronic clozapine and haloperidol on dye-coupling between neurons in the rat striatal complex. Neuroscience 66:763–767
Onn S-P, Grace AA (1995) Repeated treatment with haloperidol and clozapine exerts differential effects on dye coupling between neurons in subregions of striatum and nucleus accumbens. J Neurosci 15:7024–7036
Paxinos G, Watson C (1982) The rat brain in stereotaxic coordinates. Academic Press, New York
Reynolds GP (1992) Developments in the drug treatment of schizophrenia. Trends Pharmacol 13:116–121
Rupniak NMJ, Hall MD, Mann S, Fleminger S, Kilpatrick G, Jenner P, Marsden CD (1985) Chronic treatment with clozapine, unlike haloperidol, does not induce changes in striatal D-2 receptor function in the rat. Biochem Pharmacol 34:2755–2763
Saller CF, Salama AI (1985) Alterations in dopamine metabolism after chronic administration of haloperidol. Neuropharmacol 24:123–129
Stamford JA, Kruk ZL, Millar J (1988a) Stimulated limbic and striatal dopamine release measured by fast cyclic voltammetry: anatomical, electrochemical and pharmacological characterization. Brain Res 454:282–288
Stamford JA, Kruk ZL, Millar J (1988b) Actions of dopamine antagonists on stimulated striatal and limbic dopamine release: an in vivo voltammetric study. Br J Pharmacol 94:924–932
Stamford JA (1989) In vivo voltammetry — prospects for the next decade. Trends Neurosci 12:407–412
White FJ, Wang RY (1983) Differential effects of classical and atypical antipsychotic drugs on A9 and A10 dopamine neurons. Science 221:1054–1057
Wiedemann DJ, Garris PA, Near JA, Wightman RM (1992) Effect of chronic haloperidol treatment on stimulated synaptic overflow of dopamine in the rat striatum. J Pharm Exp Ther 261:574–579
Wilmot CA, Szczepanik AM (1989) Effects of acute and chronic treatments with clozapine and haloperidol on serotonin (5-HT2) and dopamine (D2) receptors in the rat brain. Brain Res 487:288–298
Yamada S, Yokoo H, Nishi S (1993) Chronic treatment with haloperidol modifies the sensitivity of autoreceptors that modulate dopamine release in rat striatum. Eur J Pharmacol 232:1–6
Yamamoto BK, Cooperman MA (1994) Differential effects of chronic antipsychotic drug treatment on extracellular glutamate and dopamine concentrations. J Neurosci 14:4159–4166
Youngren KD, Moghaddam B, Bunney BS, Roth RH (1994) Preferential activation of dopamine overflow in prefrontal cortex produced by chronic clozapine treatment. Neurosci Lett 165:41–44
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Feasey-Truger, K.J., Alzheimer, C. & ten Bruggencate, G. Chronic clozapine versus chronic haloperidol treatment: differential effects on electrically evoked dopamine efflux in the rat caudate putamen, but not in the nucleus accumbens. Naunyn-Schmiedeberg's Arch Pharmacol 354, 725–730 (1996). https://doi.org/10.1007/BF00166898
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00166898