Summary
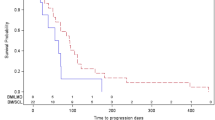
Ninety-three patients with primary intracranial brain tumors recurrent after cerebral irradiation were treated with aziridinylbenzoquinone (AZQ; Diaziquone). Twenty-four (26%) had tumor regression lasting a median of 9.2 months. Prior chemotherapy was not significantly associated with tumor regression but was associated with survival (median 7.3 months no prior chemotherapy versus 4.7 months with prior chemotherapy; logrank p = 0.03). AZQ demonstrated anti-tumor activity in a wide variety of primary intracranial neoplasms recurrent after radiation therapy and deserves study in patients at the time of diagnosis. We believe alternating or combining AZQ and BCNU should be rewarding. The principal toxicity of AZQ is myelosuppression.
Similar content being viewed by others
References
Driscoll JS, Duceck L, Congleton G, Geran RI: Potential CNS antitumor agents VI: Aziridinylbenzoquinones III. J Pharmaceut Sci 68:185–188, 1979
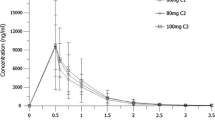
Schilsky RL, Kelley JA, Ihde DC, Howser DM, Cordes RS, Young RC: Phase I trial & pharmokinetics of AZQ (NSC 182986) in humans. Cancer Res 42:1582–1586, 1982
Eagan RT, Dinapoli RP, Hermann Jr RC, Groover RV, Layton Jr DD: Preliminary communication-treatment of primary brain tumors recurrent after irradiation with aziridinylbenzoquinone (AZQ; NSC — 182986). Am J Clin Oncol 6:577–578, 1983
Eagan RT, Scott M: Evaluation of prognostic factors in chemotherapy of recurrent brain tumors. J Clin Oncol 1:38–44, 1983
Decker DA, Al Sarraf M, Kresge C, Austin D, Wilner HI: Phase II study of aziridinylbenzoquinone (AZQ; NSC — 182986) in the treatment of malignant gliomas recurrent after radiation. J Neuro Oncol 3:9–21, 1985
Taylor SA, McCracken JD, Eyre HJ, O'Bryan RM, Neilan BA: Phase II study of aziridinylbenzoquinone (AZQ) in patients with central nervous system malignancies: a Southwest Oncology Group study. J Neuro Oncol 3:131–135, 1985
Maral J, Poisson M, Pertuiset BF, Mashaly P, Weil M, Jacquillat CL, Grillo-Loopez AJ: Phase II evaluation of diaziquone (CI-904, AZQ) in the treatment of human malignant glioma. J Neuro Oncol 3:245–249, 1985
Feun LG, Yung W-KA, Leavens ME, Burgess MA, Obbens EA, Bedikian AY, Savaraj N, Stewart DJ, Benjamin RS, Fields WS, Bodey GP: A phase II trial of 2,5-diaziridinyl 3,6-bis(carboethoxyamino) 1,4-benzoquinone (AZQ, NSC 182986) in recurrent primary brain tumors. J Neuro Oncol 2:13–17, 1984
Curt GA, Kelley JA, Kufta CV, Smith BH, Kornblith PL, Young RC, Collins JM: Phase II and pharmokinetic study of aziridinylbenzoquinone (2,5-dizairidinyl-3,6-bis(carboethoxyamino-1,4-benzoquinone, Diaziquone, NSC 182986) in high-grade gliomas. Cancer Res 43:6102–6105, 1983
Cox DR: The analysis of binary data. London, Methuen, 1970
Cox DR: Regression models and life tables. J R Statist Soc (B) 34:187–220, 1982
Kaplan EL, Meier P: Nonparametric estimation from incomplete observations. J Am Statist Assoc 53:457–481, 1958
Mantel N: Evaluation of survival data and two new rank order statistics arising in its consideration. Cancer Chemother Rep 50:163–170, 1966
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Eagan, R.T., Dinapoli, R.P., Cascino, T.L. et al. Comprehensive phase II evaluation of Aziridinylbenzoquinone (AZQ, Diaziquone) in recurrent human primary brain tumors. J Neuro-Oncol 5, 309–314 (1987). https://doi.org/10.1007/BF00148387
Issue Date:
DOI: https://doi.org/10.1007/BF00148387