Summary
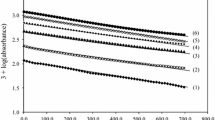
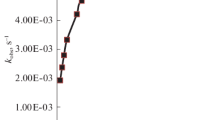
The oxidation ofDL-methionine (MT) bybis(2,2′-bipyridyl)copper(II) permanganate (BBCP) to the corresponding sulphoxide is first order in BBCP. Michaelis-Menten-type kinetics were observed with respect to MT. The formation constant of the intermediate complex and the rate constant for its decomposition were evaluated. The thermodynamic and activation parameters were also evaluated. The reaction is catalysed by H+ but 2,2′-bipyridine does not affect the reaction rate. A mechanism is proposed.
Similar content being viewed by others
References
H. Firouzabadi, M. Naderi, A. R. Sardarian and B. Vessel,Synth. Commun.,13, 611 (1983).
F. A. Walker, H. Sigel and P. McCormick,Inorg. Chem.,11, 2756 (1972).
S. Mittal, V. Sharma and K. K. Banerji,J. Chem. Res. (S), 264 (1986).
D. S. Mahadevappa, S. Ananda and N. M. M. Gowda,J. Chem. Soc., Perkin Trans.,2, 39 (1985).
S. Kothari,Proc. Indian Acad. Sci. (Chem. Sci.),103, 747 (1991).
S. Kothari, S. Sharma, P. K. Sharma and K. K. Banerji,Proc. Indian Acad. Sci. (Chem. Sci.),104, 583 (1992).
B. K. Satsangi, S. Kothari and K. K. Banerji,J. Chem. Res. (S) 208 (1994); (M) 1134.
H. Firouzbadi, A. R. Sardarian, M. Naderi and B. Vessel,Tetrahedron,40, 5001 (1984).
J. Mitchell,Organic Analysis, Interscience Publishers, New York, 1953, Vol. 1, 375.
F. Freeman and J. C. Kappos,J. Am. Chem. Soc.,107, 6628 (1985).
D. G. Lee and J. F. Perez-Benito,J. Org. Chem.,53, 5728 (1988).
K. B. Wiberg and D. Evans,J. Am. Chem. Soc.,80, 3019 (1958).
G. A. Baclay, B. F. Hoskins and C. H. L. Kennard,J. Chem. Soc., 5691 (1963).
T. Ziegler, A. Rauk and E. J. Baerends,Chem. Phys.,16, 209 (1976).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Satsangi, B.K., Kothari, S. & Banerji, K.K. Kinetics and mechanism of the oxidation of DL-methionine bybis(2,2′-bipyridyl)copper(II) permanganate. Transition Met Chem 20, 288–290 (1995). https://doi.org/10.1007/BF00143494
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00143494