Abstract
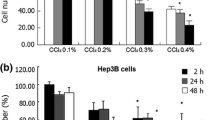
The content of reduced glutathione and of glutathione disulfide as well as the activities of glutathione reductase, glutathione peroxidase, glutathione S-transferases, catalase and superoxide dismutases were determined in human hepatoma Hep 3B cells in relation to free-radical toxicity in order to appreciate the defense capacities of these cells compared to data on normal hepatocytes. When Hep 3B cells were exposed to lindane, a known inducer of free-radical production, superoxide dismutase activity appeared as the best-adapted cellular parameter for early detection of the resulting free-radical toxicity.
Similar content being viewed by others

Abbreviations
- AAS:
-
atomic absorption spectrometry
- CDNB:
-
1-chloro-2,4-dinitrobenzene
- DMEM:
-
Dulbecco's modified Eagle medium
- GPx:
-
glutathione peroxidase
- G.Red:
-
glutathione reductase
- GSH:
-
reduced glutathione
- GSSG:
-
glutathione disulfide
- GST:
-
glutathione S-transferases
- Prot:
-
proteins
- SOD:
-
superoxide dismutase
References
Aden DP, Fogel A, Plotrin Damjanov I, Knowles BB. Controlled synthesis of HBs antigen in a differentiated human liver carcinoma derived cell line. Nature. 1979;288:615–6.
Bagchi M, Stohs SJ. In vitro induction of reactive oxygen species by 2,3,7,8-tetrachlorodibenzo-p-dioxin, endrin, and lindane in rat peritoneal macrophages, and hepatic mitochondria and microsomes. Free Rad Biol Med. 1993;14:11–18.
Baker MT, Nelson RM, Van Dyke RA. The formation of chlorobenzene and benzene by the reductive metabolism of lindane in rat liver microsomes. Arch Biochem Biophys. 1985;236(2):506–14.
Baker RD, Baker SS, LaRosa K, Whitney C, Newburger PE. Selenium regulation of glutathione peroxidase in human hepatoma cell line Hep 3B. Arch Biochem Biophys. 1993;204(1):53–7.
Bannister WH, Federici G, Heath JK, Bannister JV. Antioxidant system in tumor cells: the levels of antioxidant enzymes, ferritin, and total iron in a human hepatoma cell line. Free Rad Res Commun. 1986;1(6):361–7.
Barros SBM, Videla L, Simizu K, Van Helsema L, Junqueira VBC. Lindane-induced oxidative stress. II. Time course of changes in hepatic glutathione status. Xenobiotica. 1988;18(11):1305–10.
Bartoli GM, Galeotti T, Palombini G, Parisi G, Azzi A. Different contributions of rat liver microsomal pigments in the formation of superoxide anions hydrogen peroxide during development. Arch Biochem Biophys. 1977;184:276.
Beutler E. Effect of flavin compounds on glutathione reductase activity. J Clin Invest. 1969;48:1957–66.
Bize IB, Oberley LV, Norris MP. Superoxide dismutase and superoxide radical in the Morris hepatomas. Cancer Res. 1980;40:3686–93.
Castro VM, Söderström Carlberg I, Widersten M, Platz A, Mannervik B. Difference among human tumor cell lines expression of glutathione transferases and other glutathione-linked enzymes. Carcinogenesis. 1990;11(9):1569–76.
Chance B, Malhey AC. Assay of catalases and peroxidases. Meth Enzymol. 1955;2:764–75.
Debal A. Chimie et physico-chimie des tocophérols. In: Vitamine E tocophérols et composés apparentés. Paris: CNER-NA-CNRS Leger CL, Poytechnica Ed; 1992:1–17.
De Waziers I, Cugnenc P, Yang CS, Leroux JP, Beaune PH. Cytochrome P-450 isoenzymes, epoxide hydrolase and glutathione transferases in rat and human hepatic and extrahepatic tissues. J Pharmacol Exp Ther. 1990;253(1):387–94.
Doostdar H, Duthie SJ, Burke MD, Melvin WT, Grant MH. The influence of culture medium composition on drug metabolising enzyme activities of the human liver derived Hep G2 cell line. FEBS Lett. 1988;241(1,2):15–18.
Gad SC, Weil CS. Statistics for toxicologists. In: Hayes AW, ed. Principles and methods of toxicology. New York: Raven Press; 1989:chap 15.
Habig WH, Pabst MJ, Jakoby WB,. Glutathione S transferases. J Biol Chem. 1974;249(22):7130–9.
Halliwell B, Gutteridge JMC. Free radicals in biology and medicine. Oxford: Clarendon Press; 1985.
Hassan HM. Biosynthesis and regulation of superoxide dismutases. Free Rad Biol Med. 1988;5:377–85.
Herno V, Descampiaux B, Kuczera M, Imbenotte M, Erb F. Defence capacity modulation of human hepatoma cell line Hep 3B by three pesticides. Bull Environ Contam Toxicol. 1994;52:898–904.
Hissin P, Hilf R. A fluorimetric method for the determination of oxidized and reduced glutathione in tissues. Anal Biochem. 1976;74:214–26.
Högberg J, Kristoferson A. A correlation between glutathione levels and cellular damage in isolated hepatocytes. Eur J Biochem. 1977;74:77–82.
Junquiera VBC, Simizu K, Videla LA. Barros SBM. Dose-dependent study of the effects of acute lindane administration on rat liver superoxide anion production, antioxidant enzyme activities and lipid peroxidation. Toxicology. 1986;41:193–204.
Junquiera VBC, Simizu K, Van Halsema L, Koch OR, Barros SBM, Videla LA. Lindane-induced oxidative stress. I. Time course of changes in hepatic microsomal parameters, antioxidant enzymes, lipid peroxidative indices and morphological characteristics. Xenobiotica. 1988;18(11):1297–304.
Kuhns DB, Kaplan SS, Basford RP. Hexachlorocyclohexanes, potent stimuli of O2 - production and calcium release in human polynuclear leukocytes. Blood. 1986;68(2):535–40.
Kurihara N, Tanaka K, Nakajima M. Mercapturic acid formation from lindane in rats. Pesticide Biochem Physiol. 1979;10:137–50.
Marzin D, Hammadi R, Hautefeuille A, Vophi H. Utilisation des cellules d'hépatomes humains Hep 3B pour la réalisation du test de synthèse non programmeé de l'ADN. Forum de la société francaise de toxicologie génétique, Villejuif, 1991.
Meister A, Anderson ME. Glutathione. Annu Rev Biochem. 1983;52:711–60
Miller JN. Basic statistical methods for analytical chemistry. Part 2. Calibration and regression methods. Analyst. 1991;116:3–14.
Paglia DE, Valentine WN. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med. 1967;70:158–69.
Perini JM, Mizon C, Dehon B et al. Urinary protein determination using coomassie brilliant blue in the presence of sodium dodecyl sulphate. Clin Chim Acta. 1984;143:321–8.
Portig J, Kraus P, Stein K et al. Glutathione conjugate formation from hexachlorocyclohexane and pentachlorocyclohexene by rat liver in vitro. Xenobiotica. 1979;9(6):353–78.
Videla LA, Barros SBM, Simizu K, Junqueira VBC. Liver and biliary levels of glutathione and thiobarbituric acid reactants after acute lindane intoxication. Cell Biochem Function. 1988;6:47–52.
Videla LA. Simizu K, Barros SBM, Junqueira VBC. Mechanisms of lindane-induced hepatotoxicity: alterations of respiratory activity and sinusoidal glutathione efflux in the isolated perfused rat liver. Xenobiotica. 1991;21(8):1023–32.
Yang FL, DiSilvestro RA. Lindane induced rat liver lipid peroxidation without depressed Cu-Zn superoxide dismutase activities. Pharmacol Toxicol. 1992;70:392–3.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Descampiaux, B., Leroux, J., Peucelle, C. et al. Assay of free-radical toxicity and antioxidant effect on the Hep 3B cell line: a test survey using lindane. Cell Biol Toxicol 12, 19–28 (1996). https://doi.org/10.1007/BF00143391
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00143391