Summary
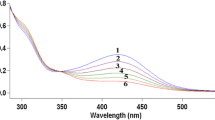
The oxidation with molecular oxygen of the cuprous salt, Cu(MeCN)4ClO4, and of bis(bromazepan)CuI perchlorate complex in the presence of different concentrations of the complexing agent bromazepan in DMSO was studied and the formation constants of the complex were calculated from the kinetic data. For comparative purposes the formation constants were also determined spectrophotometrically.
Similar content being viewed by others
References
J. Real, J. Borras, X. Solans and M. Font-Altaba, Transition Met. Chem., 12, 79 (1987) and refs cited therein.
W. Zamudio and A. M. García, Transition Met. Chem., 11, 422 (1986).
W. Zamudio and A. M. García, Transition Met. Chem., in press.
P. Hemmerich and C. Sigwart, Experentia, 19, 488 (1963).
W. Zamudio and A. M. García, Boletin Soc. Chilena de Quimica, in press.
J. Sabatino, O. Weber, G. Padmanabhan and B. Senkowski, Analyt. Chem., 41, 905 (1969).
G. Rainoni and A. Zuberbuhler, Chimia, 28, 67 (1974).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zamudio, W., Baraona, R. Synthesis, stepwise instability constants and oxidation of bis-[bromazepan]CuI perchlorate by molecular oxygen in dimethyl sulphoxide. Transition Met Chem 18, 489–492 (1993). https://doi.org/10.1007/BF00136611
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00136611