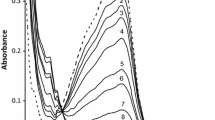
Summary
Oxidation of the FeII chelates [FeL] (L = phen or bipy) by CrVI and MnVII in H2SO4 medium was found to proceed through the formation of a bimetallic insertion complex which decomposes in the slow step, followed by electron transfer from [FeL] to the oxidant. The reactions are catalysed by both anionic and non-ionic micelles [SDS and triton-x (Tx), respectively]. A mechanism is suggested involving electrostatic stabilization of the cationic forms of the FeII chelates by anionic SDS and the partial anionic character of polyoxyethylene moiety of Tx, respectively. The marginal catalysis of cationic micelles (CTAB) is attributed to co-anion-micellar interactions.
Similar content being viewed by others
References
J. N. Ladbury and C. F. Cullis, Chem. Rev., 58, 405 (1958); J. N. Ladbury and C. F. Cullis, J. Chem. Soc., 555, 1407, 4186 (1955).
R. Stewart, Oxidation Mechanisms: Applications to Organic Chemistry, W. A. Benjamin, New York, 1964.
K. B. Wiberg, Oxidations in Organic Chemistry, Part A, Academic Press, New York, 1965
R. Stewart and M. M. Mocek, Can. J. Chem., 41, 1160 (1963).
D. Benson, Mechanisms of Oxidation by Metal Ions, Elsevier, New York, 1963, and refs cited therein.
C. H. Bamford and C. P. H. Tipper, Comprehensive Chemical Kinetics, Elsevier, New York, 1972, p. 278.
D. G. Lee and R. Stewart, J. Am. Chem. Soc., 86, 3051 (1964).
J. Rocek and J. Krupika, Collin. Czech. Commun., 23, 2068 (1958).
A. Carrington and M. C. R. Symons, Chem. Rev., 63, 443 (1963).
R. Gopalan and K. Subramanyam, J. Indian Chem. Soc., 56, 669 (1979).
N. K. Saran, M. N. Dash and R. C. Acharya, Indian J. Chem., 18A, 225 (1979).
C. A. Bunton, Nucleophilic Substitution at a Saturated Carbon Atom, Elsevier, New York, 1963.
R. S. Drago and R. E. Desimone, J. Am. Chem. Soc., 92, 2343 (1970).
A. R. J. P. Ubbelohde, J. Chem. Soc., 1605 (1935).
H. F. Launer, J. Am. Chem. Soc., 54, 2597 (1932).
W. A. Waters, Mechanisms of Oxidations, Wiley, New York, 1964; W. A. Waters, Quart. Rev., 12, 277 (1958).
V. Surender Rao, B. Sethuram and T. Navneeth Rao, Int. J. Chem. Kinet., 11, 165 (1979).
E. A. Amis, Solvent Effects of Reaction Rates and Mechanisms, Academic Press, New York, 1966.
K. C. Rajanna, G. V. Rao and P. K. Saiprakash, Z. Phys. Chem. (Leipzig), 263, 622 (1982).
H. A. Benesi and J. H. Hildebrand, J. Am. Chem. Soc., 71, 2703 (1949).
M. J. Ardon, J. Chem. Soc., 1811 (1957).
J. G. Kirkwood, J. Phys. Chem., 2, 351 (1934).
K. K. Banerji, J. Chem. Soc., Perkin Trans. 2561 (1978; K. K. Banerji, J. Chem. Soc. (M), 2561 (1978).
J. H. Fendler and E. J. Fendler, Micellar Catalysis in Organic Reactions: Kinetic and Mechanic Implication, Academic Press, New York, 1975.
C. A. Benton and G. Cerichelli, Int. Natl. J. Chem. Kinet., 11, 519 (1980).
E. Pelizetti, C. Minero, E. Pramauro and D. Meisel, J. Phys. Chem., 87, 399 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lakshmi, P.J., Rajanna, K.C. & Saiprakash, P.K. Mechanism of oxidation of iron(II) chelates by hexavalent chromium and heptavalent manganese in aqueous sulfuric acid and micellar media - a kinetic approach. Transition Met Chem 21, 135–141 (1996). https://doi.org/10.1007/BF00136543
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00136543