Summary
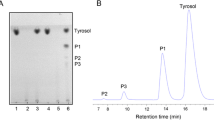
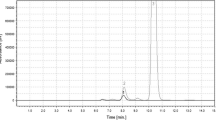
The transglycosylation activity of β-galactosidase fromAchatina achatina digestive juice was tested for glycosylating protected hydroxy amino acids. Attractive yields of β-galactosyl-(Z-Ser-OMe) (35%) and β-galactosyl-(Z-Hyp-OMe) (28%) could be obtained using lactose as glycosyl donor and the corresponding amino acid methyl esters N-protected by a benzyloxycarbonyl group (Z) as glycosyl acceptors.
Similar content being viewed by others
References
Attal, S., Bay, S., and Cantacuzene, D. (1992).Tetrahedron 48, 9251–9260.
Bay, S., Namane, A., and Cantacuzene, D. (1993).Carbohydr. Res. 248, 317–325.
Cantacuzene, D., and Attal, S. (1991a).Carbohydr. Res. 211, 327–331.
Cantacuzene, D., Attal, S. and Bay, S. (1991b).Biomed. Biochim. Acta 50, S231-S236.
Colas, B., and Attias, J. (1975).Biochimie 57, 1019–1027.
Cote, L., and Tao, B.Y. (1990).Glycoconjugate J. 7, 145–162.
Fortun, Y., and Colas, B. (1991).Biotechnol. Lett. 13, 863–866.
Holla, E.W., Schudok, M., Weber, A., and Zulauf, M. (1992).J. Carbohydr. Chem. 11, 659–663.
Johansson, E., Hedbys, L., and Larsson, P.O. (1991).Enzyme Microb. Technol. 13, 781–787.
Leparoux, S., and Colas, B. (1994).Int. J. Biochem. 26, 247–254.
Nilsson, K.G.I. (1991). Use of glycosidases and glycosyltransferases in the synthesis of complex oligosaccharides and their glycosides. In:Enzymes in carbohydrate synthesis, M.D.Bednarski and E.S.Simon, eds., pp 51–62, ACS Symposium Series 466, Washington, DC.
Sauerbrei, B., and Thiem, J. (1992).Tetrahedron Lett. 33, 201–204.
Turner, N.J., and Webberley, M.C. (1991).J.Chem.Soc.Chem.Commun. 19, 1349–1350.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Leparoux, S., Fortun, Y. & Colas, B. Synthesis of β-galactosyl-(hydroxy amino acid) derivatives using β-galactosidase activity ofAchatina achatina digestive juice. Biotechnol Lett 16, 677–682 (1994). https://doi.org/10.1007/BF00136470
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00136470