Summary
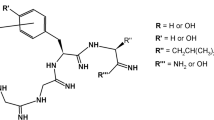
The molecular basis underlying the divergent receptor selectivity of two cyclic opioid peptides Tyr-c[N δ-d-Orn2-Gly-Phe-Leu-] (c-ORN) and [d-Pen2, l-Cys5]-enkephalinamide (c-PEN) was investigated using a molecular modeling approach. Ring closure and conformational searching procedures were used to determine low-energy cyclic backbone conformers. Following reinsertion of amino acid side chains, the narcotic alkaloid 7α-[(1R)-1-methyl-1-hydroxy-3-phenylpropyl]-6,14-endoethenotetrahydro oripavine (PEO) was used as a flexible template for bimolecular superpositions with each of the determined peptide ring conformers using the coplanarity and cocentricity of the phenolic rings as the minimum constraint. A vector space of PEO, accounting for all possible orientations for the C21-aromatic ring of PEO served as a geometrical locus for the aromatic ring of the Phe4 residue in the opioid peptides. Although a vast number of polypeptide conformations satisfied the criteria of the opiate pharmacophore, they could be grouped into three classes differing in magnitude and sign of the torsional angle values of the tyrosyl side chain. Only class III conformers for both c-ORN and c-PEN, having tyramine dihedral angles χ1 =−150° ± 30° and χ2=−155° ± 20°, had significant structural and conformational properties that were mutually compatible while respecting the PEO vector space. Comparison of these properties in the context of the divergent receptor selectivity of the studied opioid peptides suggests that the increased distortion of the peptide backbone in the closure region of c-PEN together with the pendant β,β-dimethyl group, combine to generate a steric volume which is absent in c-ORN and that may be incompatible with a restrictive topography of the μ receptor. The nature and stereo-chemistry of substituents adjacent to the closure region of the peptides could also modulate receptor selection by interacting with a charged (δ) or neutral (μ) subsite.
Similar content being viewed by others
References
A preliminary report of this work was presented at the American Crystallographic Association Annual Meeting, Philadelphia, June 26–July 1, 1988.
Simon, E.J., Hiller, J.M. and Edelman, I., Proc. Natl. Acad. Sci. U.S.A., 70 (1973) 1947.
Pert, C.B. and Snyder, S.H., Science, 179 (1973) 1011.
Goldstein, A., Science, 193 (1976) 1081.
Chang, K.J., Hazum, E. and Cuatrecasas, P., Proc. Natl. Acad. Sci. U.S.A., 78 (1981) 4141.
Duka, T.H., Schubert, P., Wüster, M., Stoiber, R. and Herz, A., Neurosci. Lett., 21 (1981) 119.
Chang, K.J., Cooper, B.R., Hazum, E. and Cuatrecasas, P., Mol. Pharmacol., 16 (1979) 91.
Chang, K.J., Hazum, E., Killian, A. and Cuatrecasas, P., Mol. Pharmacol., 20 (1981) 1.
Childers, S.R., Creese, I., Snowman, A.M. and Snyder, S.H., Eur. J. Pharmacol., 55 (1979) 11.
Portoghese, P.S., Larson, D.L., Sayre, L.M., Fries, D.S. and Takemori, A.E., J. Med. Chem., 23 (1980) 233.
Lipkowski, A.W., Tam, S.W. and Portoghese, P.S., J. Med. Chem., 29 (1986) 1222.
Fournié-Zaluski, M.C., Gacel, G., Maigret, B., Prémilat, S. and Roques, B.P., Mol. Pharmacol., 20 (1981) 484.
Roques, B.P., Fournié-Zaluski, M.C., Gacel, G., David, M., Meunier, J.C., Maigret, B. and Morgat, J.L., In Costa, E. and Trabucchi, M. (Eds.) Regulatory Peptides: From Molecular Biology to Function, Raven Press, New York, NY, 1982, pp. 321–331.
DiMaio, J. and Schiller, P.W., Proc. Natl. Acad. Sci. U.S.A., 77 (1980) 7162.
Schiller, P.W. and DiMaio, J., Nature, 297 (1982) 74.
DiMaio, J., Nguyen, T.M.D., Lemieux, C. and Schiller, P.W., J. Med. Chem., 25 (1982) 1432.
Schiller, P.W. and DiMaio, J., In Hruby, V.J. and Rich, D.H. (Eds.) Peptides: Structure and Function. (Procs. 8th Am. Pept. Symp.), Pierce Chemical Co, Rockford, IL, 1983, pp. 269–278.
Lord, J.A.H., Waterfield, A.A., Hughes, J. and Kosterlitz, H.W., Nature, 267 (1977) 495.
Sherman, D.B., Spatola, A.F., Wire, W.S., Burks, T.F., Nguyen, T.M.D. and Schiller, P.W., Biochem. Biophys. Res. Comm., 162 (1989) 1126.
Mosberg, H.I., Hurst, R., Hruby, V.J., Galligan, J.J., Burks, T.F., Gee, K. and Yamamura, H.I., Biochem. Biophys. Res. Commun., 106 (1982) 506.
Mosberg, H.I., Hurst, R., Hruby, V.J., Galligan, J.J., Burks, T.F., Gee, K. and Yamamura, H.I., Life Sci., 32 (1983) 2565.
Mosberg, H.I., Int. J. Rept. Protein Res., 29 (1987) 282.
Loew, G., Keys, C., Luke, B., Polgar, W. and Toll, L., Mol. Pharmacol., 29 (1986) 546.
Keys, C., Payne, P., Amsterdam, P., Toll, L. and Loew, G., Mol. Pharmacol., 33 (1988) 528.
DiMaio, J., Bayly, C.I., Villeneuve, G. and Michel, A., J. Med. Chem., 29 (1986) 1658.
Snyder, S.H. and Goodman, R.R., J. Neurochem., 35 (1980) 5.
DiMaio, J., Unpublished observations.
SYBYL Molecular Modelling Software, Version 5.1, Tripos Associate Inc., April, 1988.
Clark, M., Cramer, J.D. and VanOpdenbosch, N., J. Comput. Chem., 10 (1989) 982.
Marshall, G. and Kataoka, X., SYBYL Software Problem Report, 2 (1990) 2.
Gelin, B. and Karplus, M., Biochemistry, 18 (1979) 1256.
Van denHende, J.N. and Nelson, N.R., J. Am. Chem. Soc., 89 (1967) 2901.
A dummy atom is geometrically defined in space but does not contribute to the energy summations.
Kessler, H., Holzemann, G. and Zechel, C., Int. J. Pept. Protein Res., 25 (1985) 267.
Kessler, H. and Holzemann, G., Angew. Chem. Int. Ed. Engl., 20 (1981) 124.
Bentley, K.W. and Lewis, J.W., In Kosterlitz, H.W., Collier, H.O.S. and Villarieal, J.E. (Eds.) Agonist and Antagonist Actions of Narcotic Analgesic Drugs, University Park Press, Baltimore, MD, 1972, pp. 7–16.
Loew, G.H. and Perts, K., Proc. Natl. Acad. Sci. U.S.A., 75 (1978) 7.
Larson, D.L. and Portoghese, P., J. Med. Chem., 16 (1973) 195.
Michel, A.G., Proulx, M., Evrard, G., Norberg, B. and Milchert, E., Can. J. Chem., 66 (1988) 2498.
Hutchins, C.W. and Rapoport, H., J. Med. Chem., 27 (1984) 521.
Bradbury, A.F., Smyth, D.G. and Snell, C.R., Nature, 260 (1976) 165.
Portoghese, P.S., Sultana, M., Nagase, H. and Takemori, A.E., J. Med. Chem., 31 (1988) 281.
Mosberg, H.I., Hurst, R., Hruby, V.J., Gee, K., Yamamura, H.I., Galligan, J.J. and Burks, T.F., Proc. Natl. Acad. Sci. U.S.A., 80 (1983) 5871.
Zimmerman, S.S., Pottle, M.S., Némethy, G. and Scheraga, H.A., Macromolecules, 10 (1977) 1.
RMS values were calculated by SQRT (EΔ2/n−1) where n is the number of atoms within the macrocycle and Δ is the minimum distance between each atom and the middle plane.
Froimowitz, M. and Hruby, V.J., Int. J. Pept. Protein Res., 34 (1989) 88.
Hruby, V.J., Kao, L.F., Pettit, B.M. and Karplus, M., J. Am. Chem. Soc., 110 (1988) 3351.
Kerlavage, A.R., Fraser, C.M. and Venter, J.C., Trends Pharmacol. Sci., 8 (1987) 426.
Schiller, P.W., Nguyen, T.M.D., DiMaio, J. and Lemieux, C., Life Sci., 33 (Suppl. 1) (1983) 319.
Ramakrishman, K. and Portoghese, P.S., J. Med. Chem., 25 (1982) 1423.
Shiotani, S., Kometani, T., Mitsuhaski, K., Nosawa, T., Kurobe, A. and Futsukaishi, O., J. Med. Chem., 19 (1976) 803.
Freed, M.E., Potoski, J.R., Freed, E.H., Conklin, G.L. and Bell, S.C., J. Med. (Chem., 19 1976) 476.
Portoghese, P.S., Alreja, B.D. and Larson, D.L., J. Med. Chem., 24 (1981) 782.
DiMaio, J., Schiller, P.W. and Belleau, B., In Gross, E. and Meienhofer, J. (Eds.) Peptides: Structure and Function. (Procs. 6th Am. Pept. Symp.), Pierce Chemical Co., Rockford, IL, 1979, pp. 889–892.
Mammi, N.J., Hassan, M. and Goodman, M., J. Am. Chem. Soc., 107 (1985) 4008.
Hall, D. and Pavitt, N., Biopolymers, 24 (1985) 935.
Wilkes, B.C. and Schiller, P.W., Biopolymers, 26 (1987) 1431.
Belleney, J., Roques, B.P. and Fournié-Zaluski, M.C., Int. J. Pept. Protein Res., 30 (1987) 356.
Schiller, P.W., Eggimann, B., DiMaio, J., Lemieux, C. and Nguyen, T.M.D., Biochem. Biophys. Res. Comm., 101 (1981) 337.
Schwyzer, R., Biochemistry U.S.A., 25 (1986) 6335.
Schiller, P.W., Nguyen, T.M.D., Chung, N.N. and Lemieux, C., J. Med. Chem., 32 (1989) 698.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Michel, A., Villeneuve, G. & DiMaio, J. A proposal for the molecular basis of μ and δ opiate receptor differentiation based on modeling of two types of cyclic enkephalins and a narcotic alkaloid. J Computer-Aided Mol Des 5, 553–569 (1991). https://doi.org/10.1007/BF00135314
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00135314