Summary
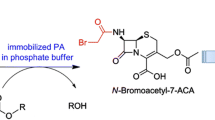
Immobilised Penicillin G acylase from E. coli hydrolyses penicillin and cephalosporin derivatives protected at the carboxy group as the phenylacetoxymethylene esters. The corresponding hydrolysis of penicillin V retains the phenoxyacetyl moiety. Kinetic data of the hydrolysis are reported.
Similar content being viewed by others
References
Baldaro, C., Fuganti, C., Servi, S., Tagliani, A. and Terreni, M. (1992), In Microbial Reagents in Organic Synthesis, S. Servi Ed., NATO ASI Series, C381 Kluwer Academic Pub. pp. 175–188
Baldaro, E., Faiardi, D., Fuganti, C., Grasselli, P. and Lazzarini, A. (1988), Tetrahedron Lett. 29, 4623
Baldaro, C., D'Arrigo, P.,Pedrocchi-Fantoni, G., Rosell, C.M., Servi, S., Tagliani, A. and Terreni, M. (1993), Tetrahedron: Asymm., 4, 1031
Fuganti, C., Grasselli, P. Servi, S., Lazzarini, A.and Casati, P., (1988), Tetrahedron 44, 2582
Fuganti, C., Rosell, C.M., Servi, S., Tagliani, A. and Terreni, M. (1992), Tetrahedron: Asymm., 3, 383
Rossi, D., Romeo, A., Lucente, G., (1978), J. Org. Chem., 43, 2576.
Rossi, D., Calcagni, A., Romeo, A., (1979), J. Org. Chem., 44, 2222.
Waldmann, H., (1991), KONTAKTE, 2, 33
Zmijeski, M.J., Briggs, B.S., Thompson, A.R. and Wright, G.I., (1991) Tetrahedron Lett. 32, 1621.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fuganti, C., Rigoni, R. & Servi, S. Selective transformations of penicillins and cephalosporins with pen G acylase. Biotechnol Lett 16, 919–922 (1994). https://doi.org/10.1007/BF00128625
Issue Date:
DOI: https://doi.org/10.1007/BF00128625