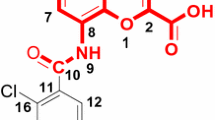
Summary
Comparative molecular field analysis (CoMFA) has been used as a three-dimensional quantitative structure-activity relationship (QSAR) method to correlate the affinities of several antagonists towards CCK-A receptors with their steric and electrostatic fields. In this publication, we describe, for the first time, a field-fit operation as an alignment technique. These results could serve as a guide for the design of new non-peptide antagonists.
Similar content being viewed by others
References
Mutt, V. and Jorpes, J.E., Klin. Wochenschr., 48 (1970) 65.
Morley, J.E., Life Sci., 30 (1982) 479.
Moran, J.H., Robinson, P., Goldrich, M.S. and McHugh, P., Brain Res., 362 (1986) 175.
Stacher, G., Psychoneuroendocrinology, 11 (1986) 39.
Mutt, V., In Glass, G.B.J. (Ed.), Gastrointestinal Hormones, Raven, New York, (1980) 169–221.
Lotti, V.J., Pendleton, R.G., Gould, R.J., Hanson, H.M., Chang, R.S.L. and Clineschmidt, B.V., J. Pharmacol. Exp. Ther., 241 (1987) 103.
Silverman, M.A., Greenberg, R.E. and Bank, S., Am. J. Gastroentero, 82 (1987) 703.
Lybrand, T., McCammon, J.A. and Wipff, G., Proc. Natl. Acad. Sci. USA, 83 (1986) 833.
Bash, P.A., Singh, U.C., Langridge, R. and Kollman, P.A., Science, 235 (1987) 574.
Goetz, M.A., Lopez, M., Monaghan, R.L., Chang, R.S.L., Lotti, V.J. and Chen, T.B., J. Antibiot., 38 (1985) 1633.
Liesch, J.M., Hensens, O.D., Springer, J.P., Chang, R.S.L. and Lotti, V.J., J. Antibiot., 38 (1985) 1638.
Chang, R.S.L., Lotti, V.J., Monaghan, R.L., Birnbaum, J., Stapley, E.O., Goetz, M.A., Albers-Schonberg, G., Patchett, A.A., Liesch, J.M., Hensens, O.D. and Springer, J.P., Science, 230 (1985) 177.
Evans, B.E., Bock, M.G., Rittle, K.E., Dipardo, R.M., Whitter, W.L., Veber, D.F., Anderson, P.S. and Freidinger, R.M., Proc. Natl. Acad. Sci. USA 83 (1986) 4918.
Evans, B.E., Bock, M.G., Rittle, K.E., Dipardo, R.M., Whitter, W.L., Veber, D.F., Anderson, P.S., Freidinger, R.M., Lundell, G.F., Chang, R.S.L., Lotti, V.J., Cerino, D.J., Chen, T.B., Kling, P.J., Kunkel, K.A., Springer, J.P. and Hirshfield, J., J. Med. Chem., 31 (1988) 2235.
Guidotti, A., Furchetti, G.M., Corda, M.G., Konkel, D., Bennet, G.D. and Costa, E., Proc. Natl. Acad. Sci. USA, 80 (1983) 3531.
Bock, M.G., Dipardo, R.M., Evans, B.E., Rittle, K.E., Whitter, W.L., Veber, D.F., Anderson, P.S. and Freidinger, R.M., J. Med. Chem., 32 (1989) 13.
Freidinger, R.M., Whitter, W.L., Gould, N.P., Holloway, M.K., Chang, R.S.L. and Lotti, V.J., J. Med. Chem., 33 (1990) 591.
SYBYL version 5.4, TRIPOS Associates, Inc., St. Louis, MO, running on Unix with Evans & Sutherland ESV III workstation.
Clark, M., Cramer III, R.D., and VanOpdenbosh, N.J., Comput. Chem., 10 (1989) 982.
Vinter, J.G., Davis, A. and Saunders, M.R.J., Comput.-Aided Mol. Design, 1 (1987) 31.
Berendsen, H.J.C., Postma, J.P.M., Van Gunsteren, W.F., Dinola, A. and Haak, J.R.J., Chem. Phys., 81 (1984) 3684.
Saunders, M.J., Am. Chem. Soc., 109 (1987) 3150.
Cramer III, R.D., Patterson, D.E. and Bunce, J.D., J. Am. Chem. Soc., 110 (1988) 5959.
Clark, M., Cramer, R.D., Jones, D.M., Patterson, D.E. and Simperath, P.E., Tetrahedron Comput. Methodol., 3 (1990) 47.
Nelder, J.A. and Mead, R., Comp. J., 7 (1965) 308.
Marshall, G.R. and Motoc, I., Topics in Molecular Pharmacology, 3 (1986) 115.
Gasteiger, J. and Marsili, M., Tetrahedron, 36 (1980) 3219.
Wold, S., Ruhe, A., Wold, H. and Dunn III, W.J., J. Sci. Stat. Comput., 5 (1984) 735.
Cramer III, R.D., Bunce, J.D., Patterson, D.E. and Frank, I.E., Quant. Struct. Act. Relat., 7 (1988) 18.
Cruse, W.B.T., Egert, E., Viswamitra, M.A. and Kennard, O., Acta Cryst., B 38 (1982) 1758.
Borea, P.A., Arnzeim.-Forsch./Drug Res., 33 (1983) 1086.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rault, S., Bureau, R., Pilo, J.C. et al. Comparative molecular field analysis of CCK-A antagonists using field-fit as an alignment technique. A convenient guide to design new CCK-A ligands. J Computer-Aided Mol Des 6, 553–568 (1992). https://doi.org/10.1007/BF00126214
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00126214