Abstract
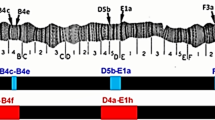
In hybrids between the sibling species D. buzzatii and D. koepferae, both sexes are more or less equally viable in the F1: However, backcross males to D. buzzatii are frequently inviable, apparently because of interspecific genetic incompatibilities that are cryptic in the F1. We have performed a genetic dissection of the effects of the X chromosome from D. koepferae. We found only two cytological regions, termed hmi-1 and hmi-2, altogether representing 9% of the whole chromosome, which when introgressed into D. buzzatii cause inviability of hybrid males. Observation of the pattern of asynapsis of polytene chromosomes (incomplete pairing, marking introgressed material) in females and segregation analyses were the technique used to infer the X chromosome regions responsible for this hybrid male inviability. The comparison of these results with those previously obtained with the same technique for hybrid male sterility in this same species pair indicate that in the X chromosome of D. koepferae there are at least seven times more regions that produce hybrid male sterility than hybrid male inviability. We have also found that the inviability brought about by the introgression of hmi-1 is suppressed by the cointrogression of two autosomal sections from D. koepferae. Apparently, these three regions conform to a system of species-specific complementary factors involved in an X-autosome interaction that, when disrupted in backcross hybrids by recombination with the genome of its sibling D. buzzatii, brings about hybrid male inviability.
Similar content being viewed by others
References
Bainbridge, S.P. & M. Bownes, 1981. Staging the metamorphosis of Drosophila melanogaster. J. Embryol. exp. Morph. 66: 57–80.
Baker, B.S. & M. Belote, 1983. Sex determination and dosage compensation in Drosophila melanogaster. Annu. Rev. Genet. 17: 345–393.
Bicudo, H.E.M.C., 1981. Nucleolar organizer activity and its regulatory mechanisms in Drosophila species of the ‘mulleri’ complex and their hybrids. Caryologia 34: 231–253.
Bock, I.R., 1984. Interspecific hybridization in the genus Drosophila. Evol. Biol. 18: 41–70.
Cabot, E.L., A.W. Davis, N.A. Johnson & C.-I. Wu, 1994. Genetics of reproductive isolation in the Drosophila simulans clade: complex epistasis underlying hybrid sterility. Genetics 137: 175–189.
Cooley, L., C. Berg & A. Spradling, 1988. Controlling P element insertional mutagenesis. TIG 4: 254–258.
Coyne, J.A., 1985. The genetic basis of Haldane's rule. Nature 314: 736–738.
Coyne, J.A., 1992. Genetics and speciation. Nature 355: 511–515.
Coyne, J.A., 1994. Haldane's rule. Nature 369: 189–190.
Coyne, J.A. & H.A. Orr, 1989. Two rules of speciation, pp. 180–207 in Speciation and Its Consequences, edited by D. Otte and J.A. Endler. Sinauer Associates, Sunderland, Mass.
Dobzhansky, Th., 1937. Genetics and the Origin of Species. Columbian University Press, N.Y.
Fontdevila, A., A. Ruiz, J. Ocaña & G. Alonso, 1982. The evolutionary history of D. buzzatii. II. How much has chromosomal polymorphism changed in colonization? Evolution 36: 843–851.
Fontdevila, A., C. Pla, E. Hasson, M. Wasserman, A. Sánchez, H. Naveira & Ruiz, 1988. Drosophila koepferae: A new member of the Drosophila serido (Diptera: Drosophilidea) superspecies taxon. Ann. Entomol. Soc. Am. 81: 380–385.
Haldane, J.B.S., 1922. Sex ratio and unisexual sterility in hybrid animals. J. Genet. 12: 101–109.
Handler, A.M., 1982. Ecdysteroid titers during pupal and adult development in Drosophila melanogaster. Dev. Biol. 93: 73–82.
Hennig, W., 1977. Gene interactions in germ cell differentiation of Drosophila, pp. 363–371 in Advances in Enzyme Regulation, Vol. 15, edited by G. Weber. Pergamon, Oxford.
Hutter, P. & M. Ashburner, 1987. Genetic rescue of inviable hybrids between Drosophila melanogaster and its sibling species. Nature 327: 331–333.
Hutter, R., J. Roote & M. Ashburner, 1990. A genetic basis for the inviability of hybrids between sibling species of Drosophila. Genetics 124: 909–920.
Kallman, K.D., 1975. The Platyfish, Xiphophorus maculatus, pp. 81–132 in Handbook of Genetics, edited by I. King, Plenum Press, NY.
Lindsley, D.L. & K.T. Tokuyasu, 1980. Spermatogenesis, pp. 226–294 in The Genetics and Biology of Drosophila, Vol. 2d, edited by M. Ashburner and T.R.F. Wright, Academic Press, NY.
Lumme, J. & E. Heikkinen, 1990. Viability of first and second generation hybrids of Drosophila virilis and Drosophila lummei. Heredity 65: 435–447.
Madhavan, K., 1973. Morphogenetic effects of juvenile hormone and juvenile hormone mimics on adult development of Drosophila. J. Insect Physiol. 19: 441–453.
Mitrofanov, V.G. & N.V. Sidorova, 1981. Genetics of the sex ratio anomaly in Drosophila hybrids of the virilis group. Theor. Appl. Genet. 59: 17–22.
Muller, H.J., 1942. Isolating mechanisms, evolution and temperature. Biol. Sym. 6: 71–125.
Naveira, H., 1992. Location of X-linked polygenic effects causing sterlity in male hybrids of Drosophila simulans and D. mauritiana. Heredity 68: 211–217.
Naveira, H. & A. Fontdevila, 1986. The evolutionary history of Drosophila buzzatii. XII. The genetic basis of sterility in hybrids between D. buzzatii and its sibling D. serido from Argentina. Genetics 144: 841–857.
Naveira, H. & A. Fontdevila, 1991a. The evolutionary history of Drosophila buzzatti. XXII. Chromosomal and genic sterility in male hybrids of D. buzzatti and D. koepferae. Heredity 66: 233–239.
Naveira, H. & A. Fontdevila, 1991b. The evolutionary history of Drosophila buzzatii. XXI. Cumulative action of multiple sterility factors on spermatogenesis in hybrids of D. buzzatii and D. koepferae. Heredity 67: 57–72.
Naveira, H., E. Hauschteck-Jungen & A. Fontdevila, 1984. Spermiogenesis of inversion heterozygotes in backcross hybrids between Drosophila buzzatii and D. serido. Genetica 65: 205–214.
Naveira, H., C. Pla & A. Fontdevila, 1986. The evolutionary history of Drosophila buzzatii. XI. A new method for cytogenetic localization based on asynapsis of polytene chromosomes in interspecific hybrids of Drosophila. Genetics 71: 199–212.
Orr, H.A., 1970. Development anomalies in Drosophila hybrids are apparently caused by loss of microchromosome. Heredity 64: 255–262.
Orr, H.A., 1993a. Haldane's rule has multiple genetic causes. Nature 361: 532–533.
Palopoli, M. & C.-I. Wu, 1994. Genetics of hybrid male sterility between Drosophila sibling species: a complex web of epistatis is revealed in interspecific studies. Genetics 138: 329–341.
Perez, D.P. & C.-I. Wu, 1995. Further characterization of the Odysseus locus of hybrid sterility in Drosophila: one gene is not enough. Genetics 140: 210–206.
Pantazidis, A.C., & E. Zouros, 1988. Location of an autosomal factor causing sterility in Drosophila mojavensis males carrying the Drosophila arizonensis Y chromosome. Heredity 60: 299–304.
Patterson, J.T. & A.B. Griffen, 1944. A genetic mechanism underlying species isolation. Univ. Texas Publ. 4415: 212–223.
Pontecorvo, G., 1943. Viability interactions between chromosomes of Drosophila melanogaster and Drosophila simulans. J. Genet. 45: 51–66.
Provine, W.B., 1991. Alfred Henry Sturtevant and crosses between Drosophila melanogaster and Drosophila simulans. Genetics 129: 1–5.
Riede, Y. & M. Renz, 1983. Study on the somatic pairing of polytene chromosomes. Chromosoma 88: 116–123.
Ruiz, A., A. Fontdevila & M. Wasserman, 1982. The evolutionary history of Drosophila buzzatii III. Cytogenetic relationship between two sibling species of the buzzatii cluster. Genetics 101: 503–518.
Sánchez, L. & A. Dübendorfer, 1983. Development of imaginal discs from lethal hybrids between Drosophila melanogaster and Drosophila mauritiana. Wilhelm Roux' Arch. Dev. Biol. 192: 48–50.
Sawamura, K., T. Taira & T.K. Watanabe, 1993a. Hybrid lethal systems in the Drosophila melanogaster species complex. I. The maternal hybrid rescue (mhr) gene of Drosophila simulans. Genetics 133: 299–305.
Sawamura, K., M.-T. Yamamoto & T.K. Watanabe, 1993b. Hybrid lethal systems in the Drosophila melanogaster species complex. II. The Zygotic hybrid rescue (Zhr) gene of D. melanogaster. Genetics 133: 307–313.
Schäfer, U., 1979. Viability in Drosophila hydei x D. neohydei hybrids and its regulation by genes located in the sex heterochromatin. Biol. Zbl. 98: 153–161.
Truman, J.W., 1971. Physiology of insect ecdysis-I. The eclosion behaviour of saturniid moths and its hormonal release. J. exp. Biol. 54: 805–814.
Watanabe, T.K., 1979. A gene that rescues the lethal hybrids between Drosophila melanogaster and D. simulans. Jpn. J. Genet. 54: 325–331.
Wharton, L., 1942. Analysis of the repleta group of Drosophila. Univ. Texas Publ. 4228: 23–52.
Wittbrodt, J., D. Adam, B. Malitschek, W. Mäueler, F. Raulf, A. Telling, S.M. Robertson & M. Schartl, 1989. Novel putative receptor tyrosine kinase encoded by the melanoma-inducing Tu locus in Xiphophorus. Nature 341: 415–421.
Wu, C.-I., & A. Beckenbach, 1983. Evidence for extensive genetic differentiation between the Sex-Ratio and the Standard arrangement of Drosophila pseudoobscura and D. persimilis and identification of hybrid sterility factors. Genetics 105: 71–86.
Wu, C.-I. & A.W. Davis, 1993. The evolution of postmating reproductive isolation: the composite nature of Haldane's rule and its genetic bases. Am. Nat. 142: 187–212.
Wu, C.-I. & M.F. Palopoli, 1994. Genetics of postmating reproductive isolation in animals. Ann. Rev. Gen. 28: 238–308.
Zeng, L.-W. & R.S. Singh, 1993. The genetic basis of Haldane's rule and the nature of asymmetric hybrid male sterility among Drosophila simulans, Drosophila mauritiana and Drosophila sechellia. Genetics 134: 251–260.
Zouros, E., 1981. The chromosomal basis of viability in interapecific hybrids between Drosophila arizonensis and D. mojavensis. Can. J. Gen. Cytol. 23: 65–72.
Zouros, E., 1989. Advances in the genetics of raproductive isolation in Drosophila. Genome 31: 211–220.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Carvajal, A.R., Gandarela, M.R. & Naveira, H.F. A three-locus system of interspecific incompatibility underlies male inviability in hybrids between Drosophila buzzatii and D. koepferae . Genetica 98, 1–19 (1996). https://doi.org/10.1007/BF00120214
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00120214