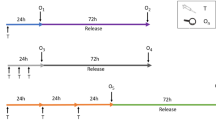
The results presented in this report demonstrate that an 18–20 hour exposure/3H-thymidine DNA labeling period is superior to a 4 hour incubation interval for general genotoxicity screening studies in the rat primary hepatocyte DNA repair assay. When DNA damaging agents which give rise to bulky-type DNA base adducts such as 2-acetylaminofluorene, aflatoxin Bi and benzidine were evaluated, little or no difference was observed between the 4 hour or an 18–20 hour exposure/labeling period. Similar results were also noted for the DNA ethylating agent diethylnitrosamine. However, when DNA damaging chemicals which produce a broader spectrum of DNA lesions were studied, differences in the amount of DNA repair as determined by autoradiographic analysis did occur. Methyl methanesulfonate and dimethylnitrosamine induced repairable DNA damage that was detected at lower dose levels with the 18–20 hour exposure/labeling period. Similar results were also observed for the DNA cross-linking agents, mitomycin C and nitrogen mustard. Ethyl methanesulfonate produced only a marginal amount of DNA repair in primary hepatocytes up to a dose level of 10−3M during the 4 hour incubation period, whereas a substantial amount of DNA repair was detectable at a dose level of 2.5 × 10−4M when the 18–20 hour exposure/labeling period was employed. The DNA alkylating agent 4-nitroquinoline-1-oxide, which creates DNA base adducts that are slowly removed from mammalian cell DNA, induced no detectable DNA repair in hepatocytes up to a toxic dose level of 2 × 10−5M with the 4 hour exposure period, whereas a marked DNA repair response was observed at 10−5M when the 18–20 hour exposure/labeling period was used.
Similar content being viewed by others
Abbreviations
- 2AAF:
-
2-acetylaminofluorene
- AB1 :
-
aflatoxin B1
- BENZ:
-
benzidine
- DEB:
-
diepoxybutane
- DEN:
-
diethylnitrosamine
- DMN:
-
dimethylnitrosamine
- EMS:
-
ethyl methanesulfonate
- MITC:
-
mitomycin C
- MMS:
-
methyl methanesulfonate
- NG:
-
mean net nuclear grain counts
- NM:
-
nitrogen mustard
- 4NQO:
-
4-nitroquinoline-N-oxide
References
BERMUDEZ, E., TILLERY, D. and BUTTERWORTH, B.E. (1979). The effect of 2, 4-diaminotoluene and isomers of dinitrotoluene on unscheduled DNA synthesis in primary rat hepatocytes. Environ. Mutagen. 1:391–398.
BRENDEL, M. and RUHLAND, A. (1984). Relationship between functional and genetic toxicology of selected DNA-damaging agents. Mutat. Res. 133:51–85.
COMBES, R.D. and HAVELAND-SMITH, R.B. (1982). A review of the genotoxicity of food, drug and cosmetic colours and other azo, triphenylmethane and xanthene dyes. Mutat. Res. 98:101–248.
CROY, R.G., ESSIGMANN, J.M., REINHOLD, V.M. and WOGAN, G.N. (1978). Identification of the principal aflatoxin B-DNA adduct formed in vivo in rat liver. Proc. Natl. Acad. Sci. USA 75:1745–1749.
DOOR, R.T., BOWDEN, G.T., ALBERTS, D.S. and LIDDIL, J.D. (1985). Interactions of mitomycin C with mammalian DNA detected by alkaline elution. Cancer Res. 45:3510–3516.
FRANCIS, A.A., SNYDER, R.D., DUNN, W.C. and REGAN, J.D. (1981). Classification of chemical agents as to their ability to induce long- or short-patch DNA repair in human cells. Mutat. Res. 83:159–169.
HALEY, T.J. (1975). Benzidine revisited: A review of the literature and problems associated with the use of benzidine and its congeners. Clin. Toxicol. 1:13–42.
HORIKAWA, M., NIKAIDO, O., TANAKA, T., NAGATA, H. and SUGAHARA, T. (1970). Carcinogenesis in tissue culture. X. Rejoining of single-strand breaks in DNA of mammalian cells induced by chemical carcinogens (4-nitroquinoline 1-oxide and its derivatives) in vitro. Exp. Cell Res. 59:147–152.
IKENAGA, M., TAKEBE, H. and ISHII, Y. (1977). Excision repair of DNA base damage in human cells treated with chemical carcinogen 4-nitroquinoline-1-oxide. Mutat. Res. 43:415–127.
IRVING, C.C. and VEAZEY, R.A. (1969). Persistent binding of 2-acetylaminofluorene to rat liver DNA in vivo and consideration of the mechanism of binding of N-hydroxy-lacetylaminofluorene to rat liver nucleic acids. Cancer Res. 19:1799–1804.
KORNBRUST, D.J. and BARFKNECHT, T.R. (1984). Comparison of rat and hamster hepatocyte primary culture/DNA repair assays. Environ. Mutagen. 6:1–11.
KRIEK, E. (1969). On the mechanism of action of carcinogenic aromatic amines. I. Binding of 2-acetylaminofluorene to rat liver nucleic acids in vivo. Chem.-Biol. Interact. 1:3–17.
LAWLEY, P.D. (1976). Carcinogenesis by alkylating agents. In: Chemical Carcinogenesis (C.E. Searle, ed.) pp. 83–244. American Chemical Society, Washington, D.C.
LOTLIKAR, P.D. (1981). Metabolic activation of aromatic amines and dialkylnitrosamines. J. Cancer Res. Clin. Oncol. 99:125–136.
MARGISON, G.P. and O'CONNOR, P.J. (1979). Nucleic acid modification by N-nitroso compounds. In: Chemical Carcinogens and DNA (P.L. Grover, ed.) pp. 111–159. CRC Press, Boca Raton, Florida.
MARTIN, C.N., BELAND, F.A., ROTH, R.W. and KADLUBAR, F.F. (1982). Covalent binding of benzidine and N-acetylbenzidine to DNA at the C-8 atom of deoxyguanosine in vivo and in vitro. Cancer Res. 42:2678–2686.
MASLANSKY, C.J. and WILLIAMS, G.M. (1982). Primary cultures and the levels of cytochrome P450 in hepatocytes from mouse, rat, hamster, and rabbit liver. In Vitro 18:683–693.
McQUEEN, C.A. and WILLIAMS, G.M. (1985). Methods and modifications of the hepatocyte primary culture/DNA repair test. In: Handbook of Carcinogen Testing (H.A. Milman and E.K. Weisburger, eds.) pp. 116–129. Noyes Publications, Park Ridge, New Jersey.
MILLER, E.C. (1978). Some current perspectives on carcinogenesis in humans and experimental animals: Presidential address. Cancer Res. 38:1479–1496.
MIRZAYANS, R. and WATERS, R. (1981). DNA damage and its repair in human normal or xeroderma pigmentosum fibroblasts treated with 4-nitroquinoline 1-oxide or its 3-methyl derivative. Carcinogenesis 2:1359–1362.
MIRZAYANS, R., PATERSON, M.C. and WATERS, R. (1985). Defective repair of a class of 4NQO-induced alkali-labile DNA lesions in xeroderma pigmentosum complimentation group A fibroblasts. Carcinogenesis 6:555–559.
MITCHELL, A.D., CASCIANO, D.A., MELTZ, M.L., ROBINSON, D.E., SAN, R.H.C., WILLIAMS, G.M. and VON HALLE, E.S. (1983). Unscheduled DNA synthesis test. A report of the U.S. Environmental Protection Agency Gene-Tox Program. Mutat. Res. 123:363–410.
MORI, H., SUGIE, S., YOSHIMI, N., KINOUCHI, T. and OHNISHI, Y. (1987). Genotoxicity of a variety of nitroarenes and other nitro compounds in DNA-repair tests with rat and mouse hepatocytes. Mutat. Res. 190:159–167.
MURNANE, J.P. and BYFIELD, J.E. (1981). Irrepairable DNA cross-links and mammalian cell lethality with bifunctional alkylating agents. Chem.-Biol. Interact. 38:75–86.
NAKAHARA, W. FUKUOKA, F. and SUGIMURA, T. (1957). Carcinogenic action of 4-nitroquinoline-1-oxide. Gann 48:129–137.
POIRIER, M.C., WILLIAMS, G.M. and YUSPA, S.H. (1980). Effects of culture conditions, cell type, and species of origin on the distribution of acetylated and deacetylated deoxyguanosine C-8 adducts of N-acetyoxy-2-acetylaminofluorene. Mol. Pharmacol. 18:581–587.
PROBST, G.S., McMAHONT, R.E., HILL, L.E., THOMPSON, C.Z., EPP, J.K. and NEAL, S.B. (1981). Chemically-induced unscheduled DNA synthesis in primary rat hepatocyte cultures: A comparison with bacterial mutagenicity using 218 compounds. Environ. Mutagen. 3:11–32.
REGAN, J.D. and SETLOW, R.B. (1974). Two forms of repair in the DNA of human cells damaged by chemical carcinogens and mutagens. Cancer Res. 34:3318–3325.
REID, T.M., WANG, C.Y., KING, C.M. and MORTON, K.D. (1984). Mutagenicity of some benzidine congeners and their N-acetylated and N, N-diacetylated derivatives in different strains of Salmonella typhimurium. Environ. Mutagen. 6:145–151.
SANCAR, A. and RUPP, W.D. (1983). A novel repair enzyme, UVRABC excision nuclease of E. coli cuts a DNA strand on both sides of the damaged region. Cell 33:249–260.
SINGER, B. (1979). N-nitroso alkylating agents: Formation and persistence of alkyl derivatives in mammalian nucleic acids as contributing factors in carcinogenesis. J. Natl. Cancer Inst. 62:1329–1339.
STARK, A.A., ESSIGMANN, J.M., DEMAIN, A.L. and WOGAN, G.N. (1979). Aflatoxin B1 mutagenesis, DNA binding, and adduct formation in Salmonella typhimurium. Proc. Natl. Acad. Sci. USA 76:1343–1347.
TADA, M. and TADA, M. (1971). Interaction of the carcinogen, 4-nitroquinoline-1-oxide, with nucleic acids: chemical degradation of the adducts. Chem.-Biol. Interact. 3:225–229.
WEISBURGER, E.K. (1981). N-substituted aryl compounds in carcinogenesis and mutagenesis. Natl. Cancer Inst. Monogr. 58:1–7.
WILLIAMS, G. M. (1976). Carcinogen-induced DNA repair in primary rat liver cell cultures: A possible screen for chemical carcinogens. Cancer Lett. 1:231–236.
WILLIAMS, G. M. (1977). Detection of chemical carcinogens by unscheduled DNA synthesis in rat liver primary cell cultures. Cancer Res. 37:1845–1851.
WILLIAMS, G.M. (1978). Further improvements in the hepatocyte primary culture DNA repair test for carcinogens: Detection of carcinogenic biphenyl derivatives. Cancer Lett. 4:69–75.
WILLIAMS, G.M., BERMUDEZ, E. and SCARAMUZZINO, D. (1977). Rat hepatocyte primary cultures. III. Improved dissociation and attachment techniques and the enhancement of survival by culture medium. In Vitro 13:809–817.
WILLIAMS, G.M., LASPIA, M.F. and DUNKEL, V.C. (1982). Reliability of the hepatocyte primary culture/DNA repair test in testing coded carcinogens and noncarcinogens. Mutat. Res. 97:359–370.
WOGON, G.N. and NEWBERNE, P.M. (1967). Dose-response characteristics of aflatoxin B1 carcinogenesis in the rat. Cancer Res. 27:2370–2376.
WOGAN, G.N., EDWARDS, G.S. and NEWBERNE, P.M. (1971). Structure-activity relationship in toxicity and carcinogenicity of aflatoxins and analogs. Cancer Res. 31:1936–1942.
YAGER, J.D. and MILLER, J.A. Jr. (1978). DNA repair in primary cultures of rat hepatocytes. Cancer Res. 38:4385–4394.
YERUSHALMI, A. and YAGIL, G. (1980). The interaction of chromatin with alkylating agents. Eur. J. Biochem. 103:237–246.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Barfknecht, T.R., Mecca, D.J. & Naismith, R.W. Effect of varying the exposure and 3H-thymidine labeling period upon the outcome of the primary hepatocyte DNA repair assay. Cell Biol Toxicol 4, 199–209 (1988). https://doi.org/10.1007/BF00119246
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00119246