Summary
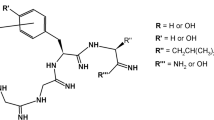
Using the method of conformational constraint, we have designed and synthesized analogues of deltorphin I containing each of the four stereoisomers of the unusual amphiphilic amino acid β-hydroxyphenylalanine in position 3. The potency and selectivity of these analogues were evaluated by radioreceptor binding assays and by bioassay in MVD and GPI. The results show that introducing a hydrophilic group into the β-carbon of Phe3 decreases the affinity and biological activity of δ-opioid receptors, which strongly depend on the chirality of the α-carbon, but not on that of the β-carbon.
Similar content being viewed by others
References
Hruby, V.J., Biopolymers, 33 (1993) 1073.
Schwyzer, R., Biopolymers (Pept. Sci.), 37 (1995) 5.
Misicka, A., Lipkowski, A.W., Horvath, R., Davis, P., Kramer, T.H., Yamamura, H.I. and Hruby, V.J., Life Sci., 51 (1992) 1025.
Misicka, A., Cavagnero, S., Horvath, R., Davis, P., Kramer, T.H., Yamamura, H.I. and Hruby, V.J., In Yanaihara, N. (Ed.) Peptide Chemistry 1992 (Proceedings of the 2nd Japan Symposium on Peptide Chemistry), ESCOM, Leiden, 1993, pp. 378–380.
Bergmann, E.D., Bendas, H. and Resmick, C., J. Chem. Soc., (1953) 2564.
Shaw, K.N.F. and Fox, S.W., J. Am. Chem. Soc., 75 (1953) 323.
Fones, W.S., J. Biol. Chem., 204 (1953) 323.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Misicka, A., Lipkowski, A.W., Stropova, D. et al. Amino acids with amphiphilic side chains: Deltorphin analogues with Phe3 replaced by all β-hydroxyphenylalanine diastereoisomers. Lett Pept Sci 2, 203–205 (1995). https://doi.org/10.1007/BF00119153
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00119153