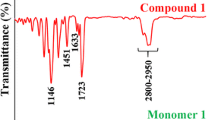
The preparation of a novel class of dimethacrylate esters, by the reaction of glycidyl methacrylate with various phthalic acid isomers and their halogen substituted derivatives is described: These monomers exhibited low viscosity, possessed similar physical and mechanical properties to bis GMA, and the presence of halogen introduced X-ray opacity into the polymer giving opacity similar to tooth enamel.
Similar content being viewed by others
References
R. L. BOWEN, US Patemt 3, 194, 783 (1965).
R. L. BOWEN, J. Dent. Res. 49 (1970) 810.
G. WILLEMS, P. LAMBRECHTS, M. BRAEM and G. VAN-HERLE, Quintessence Int. 24 (1993) 641.
K. J. M. SODERHOLM, J. Dent. Res. 62 (1983) 126.
F. F. BLIKE and F. D. SMITH, J. Amer. Chem. Soc. 51 (1929) 1865.
A. GRAHL, Ber. 28 (1895) 84.
P. J. HOLLOWAY and R. A. D. WILLIAMS, Arch. Oral Biol. 10 (1965) 237.
M. BRADEN and K. W. M. DAVY, Biomaterials 7 (1986) 474.
K. W. M. DAVY and M. BRADEN, Biomaterials 13 (1992) 1043.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Davy, K.W.M. Novel aromatic dimethacrylate esters as dental resins. J Mater Sci: Mater Med 5, 350–352 (1994). https://doi.org/10.1007/BF00058961
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00058961