Abstract
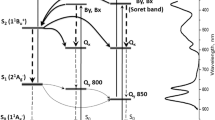
The effect of 1-hexanol on spectral properties and the processes of energy transfer of the green gliding photosynthetic bacterium Chloroflexus aurantiacus was investigated with reference to the baseplate region. On addition of 1-hexanol to a cell suspension in a concentration of one-fourth saturation, a specific change in the baseplate region was induced: that is, a bleach of the 793-nm component, and an increase in absorption of the 813-nm component. This result was also confirmed by fluorescence spectra of whole cells and isolated chlorosomes. The processes of energy transfer were affected in the overall transfer efficiency but not kinetically, indicating that 1-hexanol suppressed the flux of energy flow from the baseplate to the B806-866 complexes in the cytoplasmic membranes. The fluorescence excitation spectrum suggests a specific site of interaction between bacteriochlorophyll (BChl) c with a maximum at 771 nm in the rod elements and BChl a with a maximum at 793 nm in the baseplate, which is a funnel for a fast transfer of energy to the B806-866 complexes in the membranes. The absorption spectrum of chlorosomes was resolved to components consistently on the basis, including circular dichroism and magnetic circular dichroism spectra; besides two major BChl c forms, bands corresponding to tetramer, dimer, and monomer were also discernible, which are supposed to be intermediary components for a higher order structure. A tentative model for the antenna system of C. aurantiacus is proposed.
Similar content being viewed by others
Abbreviations
- A670:
-
a component whose absorption maximum is located at 670 nm
- (B)Chl:
-
(bacterio)chlorophyll
- CD:
-
circular dichroism
- F675:
-
a component whose emission maximum is located at 675 nm
- FMO protein:
-
Fenna-Mathews-Olson protein
- LD:
-
linear dichroism
- LH:
-
light-harvesting
- McD:
-
magnetic circular dichroism
- PS:
-
photosystem
- RC:
-
reaction center
References
Brune DC, Nozawa T and Blankenship RE (1987a) Antenna organization in green photosynthetic bacteria. I. Oligomeric bacteriochlorophyll c as a model for the 740 nm absorbing bacteriochlorophyll c in Chloroflexus aurantiacus chlorosomes. Biochemistry 26: 8644–8652
Brune DC, King GH, Infosio A, Steiner T, Thewalt MLW and Blankenship RE (1987b) Antenna organization in green photosynthetic bacteria. 2. Excitation transfer in detached and membrane-bound chlorosomes from Chloroflexus aurantiacus. Biochemistry 26: 8652–8658
Chiefari J, Griebenow K, Griebenow N, Balaban TS, Holzwarth AR and Schaffner K (1995) Models for the pigment organization in the chlorosomes of photosynthetic bacteria: Diastereoselective control of in vitro bacteriochlorophyll c s aggregation. J Phys Chem 99: 1357–1365
Feick RG and Fuller RC (1984) Topology of the photosynthetic apparatus of Chloroflexus aurantiacus. Biochemistry 23: 3693–3700
Gerola PD and Olson JM (1986) A new bacteriochlorophyll a-protein complex associated with chlorosomes of green sulfur bacteria. Biochim Biophys Acta 848: 69–76
Hanada S, Haraishi A, Shimada K and Matsuura K (1995) Isolation of Chloroflexus aurantiacus and related thermophilic phototrophic bacteria from Japanese hot springs using an improved isolation procedure. J Gen Appl Microbiol 41: 119–130
Hildebrandt P, Tamiaki H, Holzwarth AR and Schaffner K (1994) Resonance Raman spectroscopic study of metallochlorin aggregates. Implication for the supramolecular structure in chlorosomal BChl c antenna of green bacteria. J Phys Chem 98: 2192–2197
Hirota M, Moriyama T, Shimada K, Miler M, Olson JM and Matsuura K (1992) High degree of organization of bacteriochlorophyll c in chlorosomes-like aggregates spontaneously assembled in aqueous solution. Biochim Biophys Acta 1099: 271–274
Holzwarth AR, Griebenow K and Schaffner K (1990) A photosynthetic antenna system which contains a protein-free chromophore aggregate. Z Naturforsch 45c: 203–296
Kramer HJM, Pennoyer L, van Grondelle R, Westerhuis WHJ, Niederman RA and Amesz J (1984) Low temperature optical properties and pigment organization of the B875 light-harvesting bacteriochlorophyll-protens complex of purple photosynthetic bacteria. Biochim Biophys Acta 767: 335–344
Krasnovsky AA and Bystrova MI (1980) Self-assembly of chlorophyll aggregated structures. BioSystems 12: 181–194
Lutz M and van Brakel G (1988) Ground-state molecular interaction of bacteriochlorophyll c in chlorosomes of green bacteria and in model system: a resonance Raman study. In: Olson JM, Ormerod JG, Amesz J, Stackerbrandt E and Truper HG (eds) Green Photosynthetic Bacteria, pp 23–34. Plenum Press, New York and London
Matsuura K and Olson JM (1990) Reversible conversion of aggregated bacteriochlorophyll c to the monomeric form by 1-hexanol in chlorosomes from Chlorobium and Chloroflexus. Biochim Biophys Acta 1019: 233–238
Matsuura K, Hirota M, Shimada K and Mimuro M (1993) Spectral forms and orientation of bacteriochlorophylls c and a in chlorosomes of the green photosynthetic bacterium Chloroflexus aurantiacus. Photochem Photobiol 57: 92–97
Mimuro M, Nozawa T, Tamai N, Shimada K, Yamazaki I, Lin S, Knox RS, Wittmershaus BP, Brune DC and Blankenship RE (1989) Excitation energy flow in chlorosome antennas of green photosynthetic bacteria. J Phys Chem 93: 7503–7509
Mimuro M, Matsuura K, Nishimura Y, Shimada K and Yamazaki I (1992) Excitation energy transfer processes in green photosynthetic bacteria: Analysis in picosecond time domain. In: Murata N (ed) Research in Photosynthesis, Vol I, pp 17–24. Kluwer Academic Publishers, Dordrecht, The Netherlands
Mimuro M, Nozawa T, Tamai N, Nishimura Y and Yamazaki I (1994) Presence and significance of minor antenna components in the energy transfer sequence of a green photosynthetic bacterium Chloroflexus aurantiacus. FEBS Lett 340: 167–172
Mimuro M, Matsuura K, Shimada K, Nishimura Y, Yamazaki I, Kobayashi M, Wang Z-Y, and Nozawa T (1995) Molecular networks and funneling process of energy transfer in green photosynthetic bacteria. In: Mathis P (ed) Photosynthesis: From Light to Biosphere, Vol I, pp 41–46. Kluwer Academic Publishers, Dordrecht, The Netherlands
Nozawa T, Kobayashi M, Wang Z-Y, Itoh S, Iwaki M, Mimuro M and Satoh K (1995) Magnetic circular dichroism investigation on chromophores in reaction centers of Photosystem I and II of green plant photosynthesis. Spectrochim Acta 51A: 125–134
Olson JM (1980) Chlorophyll organization in green photosynthetic bacteria. Biochim Biophys Acta 594: 33–51
Olson JM and Cox RP (1991) Monomers, dimers and tetramers of 4-n-propyl-5-ethyl farnesyl bacteriochlorophyll c in dichloromethane and carbon tetrachloride. Photosynth Res 30: 35–43
Savikhin S, Zhu Y, Lin S, Blankenship RE and Struve WS (1994) Femtosecond spectroscopy of chlorosome antennas from the green photosynthetic bacterium Chloroflexus aurantiacus. J Phys Chem 98: 10322–10334
Shimada K, Yamazaki I, Tamai N and Mimuro M (1990) Excitation energy flow in a photosynthetic bacterium lacking B850: Fast energy transfer from B806 to B870 in Erythrobacter sp. strain OCh 114. Biochim Biophys Acta 1016: 266–271
van Amerongen H, Vasmel H and van Grondelle R (1988) Linear dichroism of chlorosomes from Chloroflexus aurantiacus in compressed gel and electric fields. Biophys J 54: 65–76
van Grondelle R, Sundstrom V, Dekker J and Gillbro T (1994) Energy transfer and trapping in photosynthesis. Biochim Biophys Acta 1065: 1–65
Wang Z-Y, Marx G, Umetsu M, Kobayashi M, Mimuro M and Nozawa T (1995) Morphology and spectroscopy of chlorosomes from Chlorobium tepidum by alcohol treatments. Biochim Biophys Acta 1232: 187–196
Wittmershaus BP, Brune DC and Blankenship BE (1988) Energy transfer in Chloroflexus aurantiacus: Effect of temperature and anaerobic conditions. In: Scheer H and Schneider S (eds) Photosynthetic Light Harvesting systems: Structure and Function, pp 543–554. Walter de Gruyter, Berlin
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mimuro, M., Nishimura, Y., Yamazaki, I. et al. Excitation energy transfer in the green photosynthetic bacterium Chloroflexus aurantiacus: A specific effect of 1-hexanol on the optical properties of baseplate and energy transfer processes. Photosynth Res 48, 263–270 (1996). https://doi.org/10.1007/BF00041017
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00041017