Abstract
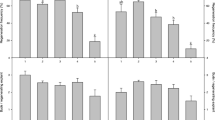
Leaf, cotyledon, and hypocotyl explants were obtained from 3-week-old seedlings of open-pollinated ‘Golden Delicious’ (Malus domestica bork H.) grown in vitro. They were placed on modified Murashige and Skoog (MS) medium containing B5 vitamins, sucrose and agar, supplemented with 6-benzylaminopurine (BAP) and α-naphthaleneacetic acid (NAA), and maintained at 25°C±2 in the light or in the dark to assess morphogenetic responses. Leaf and cotyledon explants cultured in the dark for an initial 3 weeks, then transferred to light for 4 weeks, produced 5- to 20-fold more adventitious shoots than those cultured for 7 weeks in the light. Conversely, light did not significantly influence the number of adventitious shoots formed on hypocotyl explants. Five-minute daily exposures of leaf explants to red light (651 nm) suppressed adventitious shoot formation by 80%; five-minute exposure to far-red light (729 nm) immediately following the red light counteracted the red suppression.
Seedling explants, immature fruit halves and immature embryos were also cultured on Schenk and Hildebrandt (SH) medium containing 2, 4-dichlorophenoxyacetic acid (2, 4-D), p-chlorophenoxyacetic acid (CPA) and kinetin. Light inhibited callus formation on leaf and cotyledon explants, but not on hypocotyl explants. The derived callus was placed on MS + BAP or MS + BAP + NAA for shoot regeneration. Both shoots and roots regenerated from callus placed in the dark but not in the light; the frequency of shoot regeneration was 5% or less.
Regenerated shoots were rooted on MS macronutrient salts (1/3 concentration), micronutrients, i-inositol, thiamine HCl, sucrose and agar with or without indole-3-acetic acid (IAA), indole-3-butyric acid (IBA), or NAA under a light intensity of 5.0 W.m-2 (16 h per day). Auxin concentration strongly influenced root morphology.
Similar content being viewed by others
References
Chaturvedi HC, Mitra GC (1974) Clonal propagation of citrus from somatic callus cultures. HortScience 9:118–120
Eichholtz DA, Robitaille HA, Hasegawa PM (1979) Adventive embryony in apple. HortScience 14:699–700
Gamborg OL, Miller RA, Ojima K (1968) Nutrient requirements of suspension cultures of soybean root cells. Exp. Cell Res 50:151–158
Jagger J (1977) Phototechnology and biological experiment. In: Smith KC, ed, The science of photobiology. New York: Plenum Press, pp 1–26
Kadkade P, Seibert M (1977) Phytochrome-regulated organogenesis in lettuce tissue culture. Nature 270:49–50
Karnosky DF (1981) Potential for forest tree improvement via tissue culture. BioScience 31:114–120
Krul WR, Worley JF (1977) Formation of adventitious embryos in callus cultures of ‘Seval’ a French hybrid grape. J Am Soc Hortic Sci 102:360–363
Litz RE, Conover RA (1981) In vitro polyembryony in Carica papava ovules. HortScience 16:459 (Abstr)
Liu JR, Sink KC, Dennis FG Jr (1982) Adventive embryogenesis from leaf explants of apple seedlings. (Accepted by HortScience)
Mehra A, Mehra PN (1974) Organogenesis and plantlet formation in vitro in almond. Bot Gaz 135:61–73
Milewska-Pawliczuk E, Bubicki B (1977) Induction of androgenesis in vitro in Malus domestica. Acta Hort 78:271–276
Miller CO, Skoog F (1953) Chemical control of bud formation in tobacco stem segments. Am J Bot 40:768–773
Mullins MG, Srinivasan C (1976) Somatic embryos and plantlets from an ancient clone of the grapevine (cv. Cabernet-Sauvignon) by apomixis in vitro. J. Exp Bot 27:1022–1033
Murashige T (1974) Plant propagation through tissue cultures. Ann Rev Plant Physiol 25:135–166
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assay with tobacco tissue cultures. Physiol Plant 15:473–497
Oka S, Ohyama K (1981) In vitro initiation of adventitious buds and its modification by high concentration of benzyladenine in leaf tissues of mulberry (Morus alba). Can J Bot 59:68–74
Schenk RU, Hildebrandt AC (1972) Medium and techniques for induction and growth of monocotyledonous and dicotyledonous plant cell cultures. Can J Bot 50:199–204
Skoog F, Miller CO (1957) Chemical regulation of growth and organ formation in plant tissues cultured in vitro. Symp Soc Exp Biol 11:118–130
Spiegel-Roy P, Kochba J, Dagan B (1980) Embryogenesis in citrus tissue cultures. In: Flechter A, ed, Advances in biochemical engineering, v16, Plant cell culture. Berlin: Springer-Verlag, p 27–48
Stiles HD, Biggs RH (1973) Embryoids from peach explants. HortScience 8:40 (Abstr)
Webb KJ, Street HE (1977) Morphogenesis in vitro of Pinus and Picea. Acta Hortic 78:259–269
Winton LL (1978) Morphogenesis in clonal propagation of woody plants. In: Thorpe TA, ed, Frontiers of plant tissue culture 1978. Calgary: The International Association for Plant Tissue Culture, pp 419–436
Yamaguchi T, Nakajima T (1972) Effect of abscisic acid on adventitious bud formation from cultured tissue of sweet potato. Proc Crop Sci Jpn 41:531–532
Author information
Authors and Affiliations
Additional information
Michigan Agricultural Experiment Station Journal Article No. 10695.
Rights and permissions
About this article
Cite this article
Liu, J.R., Sink, K.C. & Dennis, F.G. Plant regeneration from apple seedling explants and callus cultures. Plant Cell Tiss Organ Cult 2, 293–304 (1983). https://doi.org/10.1007/BF00039876
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00039876